Freely Available British and Irish Public Legal Information
[Home] [Databases] [World Law] [Multidatabase Search] [Help] [Feedback]
England and Wales High Court (Patents Court) Decisions
You are here: BAILII >> Databases >> England and Wales High Court (Patents Court) Decisions >> Anan Kasei Co. Ltd & Anor v Molycorp Chemicals & Oxides (Europe) Ltd [2018] EWHC 843 (Pat) (23 April 2018)
URL: http://www.bailii.org/ew/cases/EWHC/Patents/2018/843.html
Cite as: [2018] EWHC 843 (Pat)
[New search] [Printable PDF version] [Help]
BUSINESS AND PROPERTY COURTS OF ENGLAND AND WALES
INTELLECTUAL PROPERTY LIST (CHD)
Strand, London, WC2A 2LL |
||
B e f o r e :
SITTING AS A DEPUTY HIGH COURT JUDGE
____________________
| (1) ANAN KASEI CO. LTD (2) RHODIA OPERATIONS S.A.S |
Claimants |
|
| - and - |
||
| MOLYCORP CHEMICALS & OXIDES (EUROPE) LTD |
Defendant |
____________________
PIERS ACLAND QC and ADAM GAMSA (instructed by Bird & Bird LLP) for the Defendant
Hearing dates: 16, 17, 18, 19 and 24 January 2018
____________________
Crown Copyright ©
- The First Claimant (now named Solvay Special Chem Japan, Ltd.,"Solvay"), is the proprietor of European Patent No. 1 435 338 entitled "Ceric Oxide and method for production thereof and catalyst for exhaust gas clarification" (the Patent) which is designated in respect of Belgium, France, Germany, Italy and the United Kingdom. The Second Claimant Rhodia, is the exclusive licensee in respect of (at least) the German and UK designations. The Claimants are companies within the Solvay group. Solvay is a global company with headquarters in Belgium. Rhodia manufactures and sells products that are used in catalysts, including a high surface area ceric oxide for use in catalysts in vehicle exhausts. I shall call the Claimants collectively Rhodia.
- The Defendant is now named Neo Chemicals and Oxides (Europe) Ltd ("Neo"). Neo is a private limited company domiciled in the UK. It is the UK subsidiary of a global group of companies (the "Neo Group") headquartered in Toronto with corporate offices in the US and China. The Neo Group includes a Chinese subsidiary, Zibo Jiahua Advanced Materials Resource Co., Ltd ("Zibo"), which manufactures products for use in catalysts, including high surface area ceric oxide products that are used in catalysts in vehicle exhausts. Neo is responsible for the sale and distribution of these products in Europe.
- On 13 April 2016 Rhodia commenced these proceedings alleging that Neo had infringed the UK and German designations of the Patent. By Order dated 21 July 2016, Arnold J struck out the claim for relief in respect of the German designation of the Patent. Neo counterclaimed in these proceedings alleging invalidity of claims 1 to 5 inclusive of the Patent. The basis of the challenge to the validity of the Patent has changed several times since the original pleading, the current pleading being the Re-Re-Re-Re-Re-Amended Grounds of Invalidity. At trial, the grounds relied upon were obviousness of claims 1 to 5 over US 5,712,218 (Chopin), anticipation of claim 1 by the public availability of a Rhône-Poulenc product, cerium oxide 99.5 HSA-514 and insufficiency.
- Engine management systems aim to control the air/fuel mixture in a car engine to achieve a stoichiometric mixture such that the oxygen and fuel are both completely consumed eliminating the noxious gases. Under normal usage conditions, this is not possible to achieve and the conditions fluctuate from net oxidising to net reducing very rapidly, particularly as driving conditions change.
- Cerium is a 'rare-earth element" which can exist in either a trivalent (cerous) or tetravalent (ceric) state. The tetravalent ceric oxide is also known as ceria. This case is concerned with ceric oxide or ceria.
- Ceric oxide containing materials are used, and have been used for some time, in a number of different applications involving catalysis, and particularly as co-catalysts for purifying vehicle exhaust gas. They have the property of absorbing oxygen under the oxidising atmosphere and desorbing oxygen under the reducing atmosphere. This oxygen absorbing/desorbing capability of the cerium oxide containing materials enables them to purify noxious components in exhaust gases such as hydrocarbons, carbon monoxide and nitrogen oxides, by converting them to carbon dioxide and water, to carbon dioxide and to nitrogen respectively.
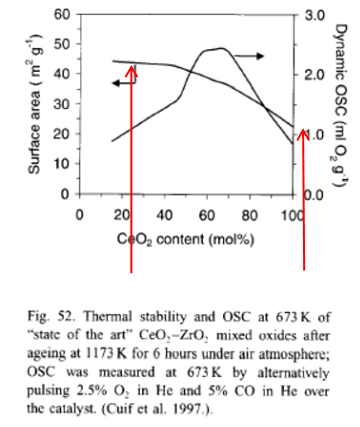
- To function efficiently in exhaust purification devices, the cerium oxide materials need to have a high specific surface area at low temperatures and to maintain a high specific surface area at the high temperatures encountered in vehicle exhaust systems. In the prior art, ceric oxide has been used in combination with other oxides, notably zirconium oxide, to stabilise its surface area at high temperatures. Without the presence of other oxides, ceria was known to sinter at high temperatures, typically 800°C and above, reducing its specific surface area and thus reducing its desirable characteristics for vehicle exhaust purification. The chart below shows the oxygen storage capacity (OSC) and the thermal stability of cerium/zirconium mixed oxides which were state of the art in 2001. The left hand arrow shows that the surface area of a 20/80% cerium zirconium mixed oxide after ageing at 1173K (900C) for 6 hours was about 45 m2/g whereas the right hand arrow shows that for a 100% ceric oxide it was about 20 m2/g.
- Ceria has also been used in a range of other applications, but none of them require it to maintain a high specific surface area at high temperatures.
- The Patent has 5 product claims, relating to specific, measurable characteristics of a ceric oxide with a high surface area. There are also 3 method claims and a product claim to a catalyst for purifying exhaust gas comprising ceric oxide of claim 1, but these are not relevant for present purposes. Infringement is alleged of claims 1 and 3 to 5. The Patent claims, in claim 1, a ceric oxide having a specific surface area of not smaller than 30.0 m2/g when subjected to calcination at 900°C for 5 hours. Calcination is essentially the process of heating to a high temperature in the presence of air or oxygen. The ceric oxide is defined as "an oxide consisting essentially of ceric oxide".
- The claim defines an obviously desirable material for use in vehicle exhaust purification devices but the material is not limited to such use. Claims 2 to 5 define the ceric oxide in terms of other specific characteristics. The specification teaches a method for making the ceric oxide of claim 1 and this is claimed in claims 6 to 8. There is no attack on the validity of these method claims. Claims 1 to 5 are not limited to products made by the process.
- There is a significant conflict between the parties as to the identity of the skilled person and this is reflected in their choices for their experts. Dr Brophy, the Defendant's expert said this:
- The Claimants' expert, Prof. Burch, agrees that the skilled person would have a background in Chemistry (most likely a graduate level qualification), would have experience in making high surface area oxides and "would be someone working in the development of high surface area oxides for use in catalytic applications, such as vehicle exhaust catalysts". He considers the Patent to be "concerned principally with those applications of ceric oxide where its oxygen storage capacity and thermal stability at high temperatures are important; namely, the application of ceric oxide in catalysts for purifying vehicle exhaust gas This high temperature stability would not generally be seen as important for the other uses of pure ceric oxide referred to in paragraph [0001]".
- In their skeleton argument the Claimants said:
- I was referred by the Claimants to the case of Schlumberger Holdings Ltd v Electromagnetic Geoservices AS [2010] RPC 33 where Jacob LJ stated in paragraph 65:
- The problem in the present case was to identify a ceric oxide material that retained high surface area at high temperatures. The only commercially important field in which this was a relevant problem was the field of automotive catalysts. People working with catalysts in other fields were not generally interested in materials retaining high surface area at high temperatures: they were only interested in high surface area in low to medium temperatures. Under cross-examination Dr Brophy was asked about the skilled person's knowledge of the maximum specific surface area that might be obtainable after calcination at 9000C and he answered:
- The Patent states in [0001] "The present invention relates to ceric oxide that has excellent heat resistance " Although it goes on to say " useful for catalysts, functional ceramics, solid electrolyte for fuel cells, and the like, " it goes on further to say " and particularly suitable for use as a co-catalyst material in catalysts for purifying vehicle exhaust gas." The only claim to a catalyst (claim 9) is one for purifying exhaust gases.
- In the light of all this, I agree with the Claimants that the skilled person is someone with a particular interest in purifying vehicle exhaust gas.
- The Claimants' expert, Professor Burch OBE, is a Fellow of the Royal Society of Chemistry and Visiting Research Professor in Catalysis at Queen's University Belfast, having previously been the Director of the Centre for the Theory and Application of Catalysis, a multicentre research initiative focussed on the application of catalytic science to industrially relevant problems. As he explains [C1/1/11] he has been involved in collaborations with industry throughout his career, including in the automotive context. He has specific experience of preparing high surface area ceric oxides.
- The skilled person is a notional construct and no real person can equate to the skilled person. Nevertheless, Professor Burch was a close approximation to the skilled person in this case and I found his evidence very useful. The Defendant had no criticism of him. He gave his evidence in a straightforward manner. In one instance he had made an error and readily accepted the fact without trying to obfuscate. He was a very good witness.
- The Defendant's expert, Dr Brophy, has a PhD from King's College and held academic posts at Leeds and Birmingham Universities prior to joining BP in 1981. At BP, he worked on the development of catalysts and catalytic processes, including for the conversion of low value hydrocarbons and natural gas to more valuable products. Dr Brophy was promoted to Technology General Manager of BP Chemicals in 1992 and worked in that role until his retirement in September 1997. While working for BP Chemicals, he also served on panels for collaborative research programs with academics and was vice-chairman of the Government's Foresight Panel for Chemicals. Following his retirement from BP, Dr Brophy worked part-time for the Royal Society of Chemistry (1997-2003), principally developing science policy and links between academia and industry. He also consults for oil and petrochemical companies in Europe, Africa and the US.
- Dr Brophy did not have the same level of knowledge in the automotive catalyst field as Professor Burch but he was clearly very knowledgeable in the wider catalyst field. The Claimants criticised Dr Brophy for his approach to the case, accusing him of "over-interpreting the literature in order to be proved right rather than an attempt to get the objectively correct outcome", and "being highly selective about the information he took as the basis for his views from the documents in the case". I agree with these criticisms. An example was the way in which Dr Brophy tended to ignore the fact that the Patent was clearly directed to ceric oxide that would maintain a high specific surface area at high temperatures, particularly suited to purifying vehicle exhaust gas and instead concentrated on the single passing reference to other applications including "functional ceramics". There is another example of selective reading to which I refer below. Although I believe that Dr Brophy was genuinely trying to help the court I did find that he was too prepared to argue his client's case rather than reading the documents with an open mind.
- I found his evidence useful but where there is a conflict with Professor Burch I prefer the evidence of Professor Burch.
- Dr Gilbert Blanchard was called and cross examined concerning the supply of catalysis products, particularly cerium containing materials for the automobile emission control, to research institutions prior to the priority date of the Patent, which products were relied on by Neo as a prior publication of the alleged invention Dr Blanchard gave evidence with the assistance of an interpreter, his native language being French.
- I found him to be a good witness doing his best to help the Court. He was being asked about events in the 1990s and, naturally, had some difficulty remembering precise details. No criticism was made of Dr Blanchard. In the event Dr Blanchard's evidence was not relevant as Neo did not pursue the anticipation attack.
- Because of the differences between the parties over the identity of the skilled person, as outlined above, there were differences as to the common general knowledge and particularly the sources to which the skilled person would refer. Thus, Dr Brophy included the Journal of the American Ceramic Society, the Journal of the European Ceramic Society and the Journal of Materials Chemistry. Similarly, whilst Professor Burch identifies conference proceedings that would have been of interest to the Skilled Person: Catalysis and Automotive Pollution Control; International Congress on Catalysis; International Conference on Environmental Catalysis; Society of Automotive Engineers conference series, Dr Brophy opines that the Skilled Person would "be aware of (and may have attended) conferences which were relevant to the particular sector or application in which he was working for example advanced materials, fuel cells and catalysts for refining and petrochemicals"
- The Claimants put to Dr Brophy some of the evidence he had given in the earlier case of CompactGTL Limited v Velocys, in which he was engaged as an expert witness, and he agreed with it in this case. The evidence was as follows:
- This accords very well with Professor Burch's view. I do not accept that the skilled person in this case would have consulted the journals referred to by Dr Brophy, nor do I accept that the skilled person would have been particularly interested in conferences relevant to the sectors identified by Dr Brophy unless there was a specific topic concerning heat stability of catalysts.
- I find that the following were part of the common general knowledge:
- When a metal salt is dissolved in water it will hydrolyse to produce H+ or H3O+ ions and metal hydroxide. This will raise the acidity of the solution. When attempting to measure the pH of the solution by, for instance, alkalimetric titration, which involves the neutralisation of the H+ ions by the measured addition of a base, the neutralised H+ ions can be replaced by H+ ions generated by the hydrolysis reaction giving an overestimate of the concentration of H+ ions in solution. The term free acidity was used in the prior art document relied on, US patent No 5712218 ("Chopin"), in connection with accounting for this effect. The experts agreed that this term was common general knowledge but disagreed as to the meaning of it. The meaning is important as Dr Brophy used his understanding of the term as an important plank in his obviousness argument based on the Chopin prior art. I shall deal with this disagreement when addressing the arguments on obviousness over Chopin below.
- The priority date of the Patent is 7 September 2001.
- Having recited the prior art the Patent defines the object of the claimed invention:
- When cross-examined about the technical contribution of the Patent, Dr Brophy gave the following answers:
- Claim 1 of the Patent is the main product claim: A ceric oxide which is an oxide consisting essentially of ceric oxide, and wherein said ceric oxide has a specific surface area of not smaller than 30.0 m2/g when subjected to calcination at 900°C for 5 hours.
- There is an issue as to the meaning to be given to "consisting essentially of ceric oxide" which is said by the Defendant to be ambiguous and leads to insufficiency which I will deal with below.
- Claims 2 to 5 relate to ceric oxide products having certain additional characteristics.
- Claim 6 is the main method claim: A method for preparing a ceric oxide of claim 1, comprising the steps of:
- There is a dispute between the parties' experts as to the characterisation of this process. Dr Brophy's evidence is that the Skilled Person would recognise it to be a hydrothermal synthesis because it involves heating the cerium solution. Professor Burch does not consider it to be a hydrothermal synthesis because that would produce a precipitate after the heating step at (b) whereas the Patent describes the production of a precipitate only after the addition of base at (d). This is significant when considering the obviousness attack dealt with below.
- Example 1 is in accordance with the method of the invention:
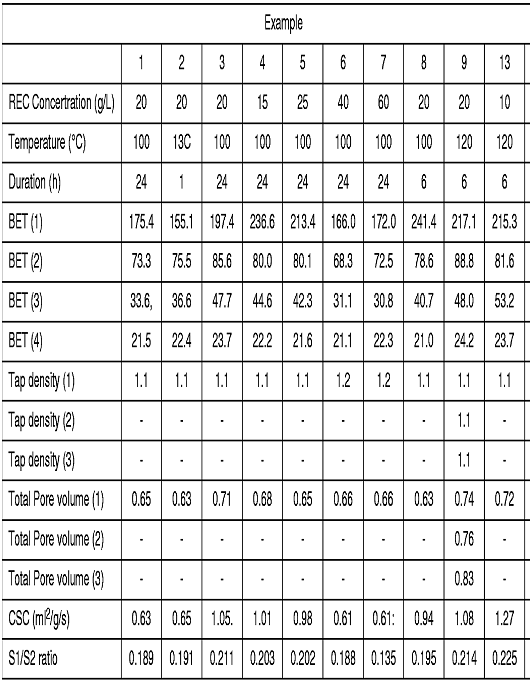
- Further examples were carried out with the following results, except that there is a misprint and Example 13 should be Example 10:
- The BET(3) results are measurements of the surface area and correspond to measurements taken after the calcination at 900° C for 5 hours test required by claim 1. All of the examples are over the bottom limit required by claim 1 to varying degrees.
- The inventive concept of the invention is the production of a ceria product that maintains a high specific surface area after subjected to high temperatures.
- Neo pleaded a case of anticipation by the supply of a ceric oxide material by Rhône-Poulenc, the predecessor of the Rhodia group. Neo relied upon the publication of a paper entitled "Interactions of CO with Pt/ceria catalysts" as evidence that the product was freely available on the market before the priority date of the Patent. The evidence of Dr Gilbert Blanchard was directed to the availability of this product and the conditions of confidentiality attached to its supply. Rhodia located three containers of the material and these were analysed and were claimed by Neo to be within claim 1 of the Patent. This was disputed by Rhodia. This turned on the proper construction to be given to the qualification in claim 1 "consisting essentially of ceric oxide".
- In closing Neo submitted that the claims are insufficient on Rhodia's construction and did not rely on the anticipation allegation.
- At paragraph 23 of Pozzoli, Jacob LJ reformulated the Windsurfing approach as follows:
- I have already identified the relevant skilled person and the common general knowledge. I have also identified the inventive concept of the claim.
- Neo does not allege obviousness over the Rhône-Poulenc material but does allege obviousness over US Patent No. 5,712,218 published on 27 January 1998 ("Chopin") together with common general knowledge. Chopin is owned by Rhône-Poulenc and Neo contends that it discloses the same method as the Patent which is an attempt to 'evergreen' its hydrothermal synthesis method of preparing rare earth oxides.
- Chopin is stated to be directed to "Cerium/Zirconium mixed oxide catalysts having high/stable specific surface areas". In column 1, lines 20 to 25 it states:
- In the Summary of the Invention at column 2, lines 30 to 41 Chopin identifies the objects of the invention:
- Two things are apparent from these passages, namely: (1) that the invention is aimed at producing mixed oxides and not pure ceric oxide; and, (2) that the invention is aimed at producing the mixed oxides which retain a large specific surface area after calcination at elevated temperatures.
- In column 2, lines 8 to 26, Chopin describes how normally the preparation of a solid solution requires calcination at high temperature which leads to a loss in specific surface area.
- The summary of the method is described at column 3, lines 18 to 29:
- Dr Brophy considers this to be a hydrothermal synthesis. Professor Burch originally described it as a co-precipitation, however he subsequently accepted that he had misread the document and accepted that it is a hydrothermal synthesis.
- Column 3, lines 39 to 57 say that the resulting product preferably has a specific surface area of at least 150 m2/g and will maintain a specific surface area of preferably 30 m2/g after calcination at 900°C. Dr Brophy considers that the skilled person would find these values of great interest for a range of applications.
- Column 3, lines 60 onwards describe the x-ray diffraction analyses which show zirconia and ceria co-existing in a single identifiable phase for a range of compositions for CexZr(1-x)O2 from x = 0.4 to 1 (excluding the latter value). 100% ceric oxide is therefore excluded. Professor Burch accepted in cross-examination that the formula could be read as excluding only vanishingly small quantities of zirconia. As a matter of mathematics, it plainly does. As 100% pure ceria does not exist, the formula would be understood by the skilled person to include pure ceria. However, Professor Burch's evidence was that, taken as a whole, Chopin is clearly directed to mixed oxides and not pure ceria.
- A more detailed description of the method of synthesis is described from column 4, line 35 onwards.
- The first stage of the process entails preparing an aqueous mixture of metal salts, for example by dissolving soluble compounds in water, or by mixing solutions of the same. Exemplary cerium (IV) compounds are nitrates or ceric ammonium nitrate (often referred to as CAN). The patentee's preference is to use ceric (IV) nitrate containing at least 85% cerium (IV) and various methods of producing that nitrate are disclosed including aqueous solutions and oxidation of trivalent cerium (including electrolytic oxidation).
- Column 4, line 61 onwards refers to the initial free acidity of the aqueous solution of the cerium (IV) salt as being from 0.1 to 4 N. This initial free acidity may be neutralised by the addition of a base and a formula is provided for achieving the required degree of neutralisation.
- Column 5, line 38 onwards describes the second stage in the synthesis which involves heating the solution in a "thermohydrolysis" step. The stated temperature range is 80 to 350°C for 1 to 48 hours.
- Column 6, line 13 describes at the end of the heating, recovering a solid product by standard separation techniques e.g. filtration, separation, draining or centrifugation.
- Three optional steps are described:
- Column 6, line 60 to column 7, line 8 describes a list of applications of the mixed oxide product made by the method of Chopin. Several of those, albeit not all, were known applications of ceria or applications for which the use of ceria was the subject of research.
- Five illustrative examples are disclosed, together with a single comparative example.
- Dr Brophy explains that:
- At column 7 lines 52 to 61, Chopin compares X-ray diffraction spectrum of a "pure ceric oxide prepared under the same conditions" as used in Example 1 but in the absence of zirconium with the X-ray diffraction spectrum of the product of Example 1. This is said to establish that the product of Example 1 has the zirconium incorporated into the crystalline lattice of the cerium oxide by the displacement in the position of the diffraction peaks with no detectable peaks from a secondary phase.
- Although Chopin discloses the production of pure ceria by the method used to produce mixed oxides, it is only for the purpose of proving the crystal structure of the mixed oxides. There is no investigation of the properties of the pure ceria that was produced with the implication, as suggested by Professor Burch, that the material is of poor quality and of no further interest from the view of thermal stability although he could not put forward any technical basis to support that implication. Although the passage in Chopin describing the range of mixed oxides produced according to the invention could be read as including pure ceria, I accept Professor Burch's evidence that the skilled person would not read it in that way in the overall context of the specification which is directed to mixed oxides with a minimum of 10% of other oxides.
- Neo rely on Dr Brophy's evidence that it would be obvious to synthesise pure ceria using the method of Example 2 of Chopin which makes the best mixed oxide product in terms of surface area, 39m2/g after calcination at 900°C for 6 hours. If this were to be achieved with ceria rather than with a mixed oxide of cerium, zirconium and yttrium, this would be within claim 1 of the Patent.
- The attraction of the Chopin method to the skilled person, according to Dr Brophy, is that it is a simple, low temperature, and therefore a potentially low cost method. Dr Brophy would expect that it could be used for the preparation of high surface area pure ceria at low cost. No experiments were performed to confirm that this would be so. Instead, Neo argue that the obvious way for the skilled person to perform the Chopin method would be the same as the method disclosed and claimed in the Patent. In those circumstances, Neo argue, either the result is within claim 1 of the Patent or the Patent is insufficient.
- I must therefore look at the Chopin method and particularly the way in which Dr Brophy says that it would be of interest to the skilled person interested in producing ceria and the way in which it would be obvious to the skilled person to implement it.
- However, there is a preliminary point taken by Rhodia as to the method by which Dr Brophy's evidence on this was obtained. Dr Brophy's evidence as to the method that would have been used by the skilled person was in his Further Expanded Report. By the time that he produced that Report, Dr Brophy was very familiar with the Patent and, in fact, it appears that he had the Patent in front of him when he prepared it. Therefore, Rhodia's submission is that Dr Brophy was inevitably using hindsight when he prepared this evidence. Dr Brophy stated that he had been instructed to "put (the Patent) out of the picture" but his evidence was in the form of a comparison between the conditions disclosed in Chopin and the conditions set out in the Patent. He then concludes that the conditions that would be used by the skilled person would fall within the ranges specified in the Patent.
- This method of dealing with this issue is unfortunate and clearly makes it difficult for the evidence not to be tainted by hindsight. The evidence would be fine if Dr Brophy had been able to say that he had looked at Chopin first and had identified the conditions that he believed the skilled person would have adopted to make ceria based on the teaching of Chopin and common general knowledge, but he was unable to say that. This is not the fault of Dr Brophy but is an inevitable result of the late amendments to Neo's case on invalidity. I accept Rhodia's criticisms of this evidence and, although I do not reject Dr Brophy's evidence on this issue I can give it little weight.
- As I stated above, Dr Brophy was inclined to read only the parts of documents that supported his client's arguments and ignored anything that taught against them. An example of this was his justification for the skilled person to be interested in the hydrothermal synthesis of Chopin for the production of a pure ceria product rather than a mixed oxide product. He produced a list of the advantages of hydrothermal synthesis taken from a table in a paper by Somiya and Roy and which he stated were "well known". However, Dr Brophy left out one item from the table which was that powders produced by hydrothermal synthesis are "highly reactive in sintering". I would add this reactivity of powders in sintering, when produced by hydrothermal synthesis, to the list of common general knowledge.
- Dr Brophy justified the omission because he was talking about the attraction of hydrothermal synthesis to "a whole raft of people who are interested in low temperature applications". In cross-examination he accepted that it would be highly relevant to auto exhaust systems because sinterability is exactly what you don't want. Indeed, the reason for using mixed oxides rather than pure ceria is their greater resistance to sintering. I find that the skilled person in the automotive field would have little interest in Chopin for producing pure ceria product rather than a mixed oxide product.
- Neo's attack on obviousness is based on "the whole raft of people who are interested in low temperature applications" who are looking for different methods or producing ceria with high surface area at low temperatures. These are people who are not interested in high temperature characteristics but just want a low cost method for producing high surface area ceria at low temperatures and would never take the product above 8000C. If they did produce ceria with the high temperature surface areas of the Patent, and therefore within at least claim 1 of the Patent, they would never know they had done it. Neo says that doesn't matter: if it was obvious for them to obtain the product even without knowing they had done so, the claim would be obvious. Neo relies on the statement of the law in Napp v ratiopharm [2009] EWCA Civ 252 by Jacob LJ:
- Applying that here, if it would be obvious to someone in the general catalysis field to produce ceria by the Chopin method and they produced a pure ceria that had the extra advantage of higher surface area at high temperatures that would make the claims obvious. It is not necessary that he should expect or intend to obtain that advantage or even know that he had.
- Thus, although the skilled addressee of the Patent would be put off from trying Chopin to produce pure ceria because he or she would expect that ceria to be highly reactive in sintering I have to consider whether someone more generally interested in low temperature catalysts would find it obvious to make a ceria product falling within claim 1 of the Patent by implementing Chopin.
- This person will not be put off by the risk of the pure ceria product being highly reactive in sintering. On the other hand, he/she might be put off by the fact that Chopin is directed to mixed oxides for use in high temperature environments. Dr Brophy suggests that such a person would be interested in finding a low temperature, low cost method of production for ceria. However, I prefer Professor Burch's evidence that this would not be seen as disclosing an interesting new method for preparing a pure ceria product but would be seen as directed to producing improved mixed oxides and therefore of no particular interest to someone wanting to prepare a pure ceria product. It is Dr Brophy's evidence that thermal hydrolysis was well known as was demonstrated by his reference to the Somiya and Roy paper referred to above so the method of production would not be seen as anything unusual.
- There is a difficulty with performing Example 2 of Chopin in that there is no direct statement of the starting concentration of the ceric nitrate. Example 2 refers to "a solution of ceric nitrate having a free acidity of 0.62N". Dr Brophy calculates this to indicate a concentration of 26.7 g/l of ceric nitrate, which is within the range of concentrations used in the examples in the Patent but this is not accepted by Professor Burch. The calculation appears to be based on the assumption that all the acidity referred to as "free acidity" comes from the hydrolysis of the Ce4+ ions. Thus his calculation assumes that all of the ceric nitrate is hydrolysed and that 4 protons are produced by the hydrolysis of each Ce4+ ion.
- As I stated above, although the experts both agree that "free acidity" is part of the common general knowledge, there is a disagreement between them as to what the meaning of the term is. Professor Burch states that free acidity is the acidity of a solution excluding acidity produced by hydrolysis of hydrolysable ions and therefore would give no indication as to the concentration of ceric nitrate in the starting solution. In his written evidence, Dr Brophy seemed to be saying that he understood free acidity to exclude any additional acidity due to further hydrolysis of cationic cerium but to include any acidity that has resulted from hydrolysis prior to the titration. At the start of his cross-examination, Dr Brophy agreed that his position was that "free acidity can include a component from the acidity that comes from the hydrolysis of metal ions prior to the titration". However, later in his cross-examination Dr Brophy said that in measuring the free acidity you are trying to measure "the original acidity that was in the solution before you added the cations".
- Dr Brophy's evidence in his first report was that ceric salts hydrolyse readily in solution and a freshly made solution would ultimately form a colloidal suspension or a precipitate. To stabilise ceric nitrate some nitric acid can be left in the synthesised crystals or added to the solution to keep it in solution. When the solution is used therefore, it will have an appreciable acidity which may need to be neutralised. Although he attempted to go back on this evidence in cross-examination, he accepted "If you buy a solution of ceric nitrate, it usually contains a lot of acid. It has to otherwise it would hydrolyse like the clappers and you would end up with particles on the bottom."
- Chopin explains that free acidity can be neutralised according to the equation for the ratio of neutralisation given at column 5, line 7:
- Neo sought to counter this in closing by producing a rather complicated version of the formula from column 5 of Chopin, taking into account the various species of cerium present, which is said to be consistent with the explanation in column 5 under the formula. It is then said that because Examples 1 and 3 include an instruction to adjust the starting solution of ceric nitrate using that formula and Example 2 does not include such an instruction, Example 2 would be understood by the skilled person to use a starting solution of ceric nitrate dissolved in water, with no acid, the equation for the ratio of neutralisation is inappropriate. I cannot accept that this is a correct interpretation of Chopin. It is not consistent with Neo's opening skeleton, nor is it consistent with Dr Brophy's evidence, referred to above, that ceric nitrate crystals would be supplied with nitric acid present in them.
- On the basis of this equation in Chopin, Professor Burch's evidence as to the meaning of free acidity in the common general knowledge and the fact that ceric nitrate crystals would be supplied with nitric acid in them, the identification of the free acidity of the starting solution of ceric nitrate in Example 2 of Chopin cannot be used to calculate the starting concentration of ceric nitrate in that example.
- The solutions in the Patent examples range from 20-60 g/L with 40 g/L and 60 g/L being only just above the 30.0 m2/g lower limit for the specific surface area in claim 1. Professor Burch gave evidence that the skilled person would expect lower concentrations to give larger particle sizes and lower surface areas than more concentrated solutions. This would teach against using the concentrations used in the examples of the Patent and I prefer Professor Burch's evidence to Dr Brophy's suggestion that the skilled person would use a dilute concentration as used in the examples in the Patent when trying to carry out Example 2 of Chopin.
- I will not go through the choice of other parameters where Chopin sets out a range and Dr Brophy selects a parameter from that range which is within the range set out in the Patent as these choices are tainted by hindsight as I have stated above.
- Thus the main differences between the inventive concept of the Patent and the disclosure of Chopin are:
- It would not have been obvious to the skilled addressee, whether in the automotive catalysis field or the more general catalysis field to take Chopin as a starting point, to ignore its teaching relating to mixed oxides and to use it to make pure ceria. Even if the skilled person were to try making ceria by the method of Chopin, it would not be obvious to that skilled person to adapt the method of Chopin so as to achieve a product within claim 1 of the Patent.
- Neo claims that "the specification does not contain any directions or explanation as to the meaning of the term "consisting essentially of ceric oxide". In the premises, the skilled person would be unable to implement the invention or determine whether he was working the same without undue effort or at all."
- Rhodia relies on a settled line of European Patent Office Boards of Appeal case-law to the effect that "consisting essentially of" means that apart from the components which are mandatory (i.e. the ceric oxide), no other components may be present in the composition which materially affect the essential characteristics of the composition.
- This line of case-law is referred to in the Boards of Appeal case-law book 2016 edition at page 288, and is most conveniently summarised in §1.2.3 of Case T 1730/09 as follows (original emphasis):
- Rhodia argues, correctly, that the Court is entitled to take this convention into account when construing the claims of the Patent through the eyes of the skilled person see Virgin Atlantic v Premium Aircraft [2009] EWCA Civ 1062 at [13]. Rhodia goes on to say it would be wrong for the Court to depart from such a settled approach in the EPO see the observations of the Court of Appeal in Actavis v Merck [2008] RPC 26 at §§44-48 and 93-107 and also reflected in the decisions of the House of Lords and Supreme Court in Merrell Dow v Norton, Conor v Angiotech, Generics v Lundbeck and Lilly v HGS and that, in any event, the same approach is adopted in the UK Manual of Patent Practice see §14.123.1 which recites the above EPO case-law and explains "[t]his settled view of the EPO is followed in the UK."
- Neo submits that the language is ambiguous with the consequence that all of the claims of the Patent are invalid for insufficiency. It differentiates a case where it is possible to postulate difficult questions about a borderline, a "fuzzy boundary" and a specification which makes it impossible for the skilled person to know whether a product falls within the claim because he is uncertain as to what criterion to use.
- Neo relies on the leading authority in this respect Kirin-Amgen v Hoechst [2004] UKHL 46, [2005] 1 All ER 667. In that case, claim 19 of the patent was for recombinant erythropoietic (rEPO) characterised in having a higher molecular weight by SDS-PAGE that erythropoietin isolated from urinary sources (uEPO). SDS-PAGE is and was a well-known method of ascertaining the apparent molecular weight of proteins.
- The evidence at trial was that the molecular weight of uEPO varied. Such variations might have been attributable to the source of the urine and the method of purification or might have been purely random. However, the patent did not identify the uEPO which was to be used as the comparator. The House of Lords held that claim 19 was invalid for insufficiency. As Lord Hoffmann explained at [124]-[126]:
- Neo points out that whilst there is a settled approach in the EPO as to the meaning of "consisting essentially of", in none of the decisions following that approach has the Board of Appeal sought to address the practical implications of this approach. This is understandable as decisions on infringement are not within the scope of their jurisdiction.
- I believe that there are two issues here. The first is the issue of construction as to what does "consisting essentially of" mean, and the second is whether that is sufficient on the facts of this case.
- As to the first issue, I believe that I should follow the settled approach of the EPO and construe "consisting essentially of" as meaning that apart from the components which are mandatory (i.e. the ceric oxide), no other components may be present in the composition which materially affect the essential characteristics of the composition.
- Neo points out some of the practical problems with that construction in the present case. Claim 1 is a product claim and it must be possible for the skilled person to ascertain from the product alone whether it satisfies the requirements of the claim. The specification identifies the relevant threshold surface area required as not less than 30m2/g after calcination at 900oC for 5 hours and the experimental technique to be used to measure it. Clearly that causes no problems. The skilled person can use the experimental technique to measure the surface area after the specified calcination and determine whether it is above the required minimum.
- However, it is also necessary to determine whether the product "consists essentially of ceric oxide". Professor Burch identified two techniques for analysing the composition of the product although neither is capable of analysing certain elements, including carbon. Having carried out the analysis, the skilled person would know the composition of the product, at least in respect of most elements in the periodic table except for carbon. To know whether the product consists essentially of ceric oxide it is necessary for the skilled person to know the effect that each of the 'impurities' in the ceric oxide would have on the surface area.
- Neo argues that this poses a problem since, for instance, it is known that a mixed oxide containing zirconium may affect the surface area after calcination, depending on the amount present. It is not known how much zirconium will have a material effect on the surface area of the product. Neo says that the effect will depend on the amount present and on the method of producing the product. A purchaser of the product will not know how it has been made. They may carry out the analysis and determine that there is some zirconium present but, if it is a small amount, they will not know whether it has materially affected the surface area of the product. I believe that this is a "fuzzy boundary" and is not a true insufficiency. There is often a limit at the edge of a claim where the precise limit is difficult to ascertain. That does not lead to a finding of invalidity.
- There is a further problem in that the common general knowledge methods of analysis are unable to detect carbon. It was accepted by Professor Burch that organic (i.e. containing carbon) surfactants can also affect the thermal stability of the product. The skilled person would be unable to determine whether or not a surfactant had been included in the product by analysis unless they knew the details of its manufacture. Someone purchasing the product could not tell.
- I accept that this could be a problem however, I do not believe that the skilled person would believe that the patentee had intended to exclude the use of a surfactant in the production of the product. The Patent is clearly directed to producing ceric oxide in place of the mixed oxides being used at the priority date of the Patent. I find that this head of the insufficiency attack fails.
- Neo submits that this is a case where the breadth of the claim exceeds the technical contribution to the art made by the invention and is therefore insufficient in accordance with the principles set out by Lord Hoffmann in Biogen v Medeva [1997] R.P.C. 49. Arnold J summarised the law in Sandvil v Kennametal [2012] RPC 23 as follows:
- In this case, Neo submits that the claims of the Patent are not to a single product, such as the enantiomer in Lundbeck, but to a multitude of products with different structures and characteristics, depending on the method of synthesis used. The claim, says Neo, is readily distinguishable from that in Lundbeck. Neo submits that claims 1-5 of the Patent are framed simply by reference to known desirable properties of a product what is sometimes called a 'free beer' claim. Neo relies on the following passage in the judgment of Jacob LJ in Lundbeck v Generics [2008] EWCA Civ 311 which were obiter and not considered by the House of Lords:
- Neo says ceric oxide was a known product and the skilled person appreciated that good thermal stability was a desirable characteristic of ceric oxide. The Patent therefore claims a desirable result. Moreover, it claims all forms of ceric oxide with that physical characteristic while only enabling a subset, namely the forms of ceric oxide produced by the method taught in the Patent. The skilled person would know that different methods of manufacture (including, for example, the use of surfactants) could produce ceric oxide with different morphologies (i.e. different physical structures).
- In the present case the claims are to ceric oxide having certain properties. The Patent teaches a range of conditions to achieve a product with the favourable characteristics claimed. Dr Brophy accepted in cross-examination when it was put to him that the reader is taught a general principle which can then be applied, answering "I think, in this particular case, yes". This case can be distinguished from Biogen and is in line with the situation in Lundbeck. I find that this insufficiency attack fails.
- Neo's objection is that the numerical limit in claim 1 is unbounded in that there is no stated upper limit to the surface area of the product but the Patent does not teach infinitely high surface area after the calcination.
- Professor Burch stated that it would be obvious to a skilled person reading the Patent that there is a practical upper limit and that not all values above 30 m2/g would be achievable. Dr Brophy agreed.
- Rhodia's answer is that the skilled person would be able to identify the upper limit enabled by the teaching of the Patent by routine trial and error. I agree. I do not think that this is a case, as alleged by Neo, that the claims of the Patent exceed its technical contribution. This insufficiency attack also fails.
- Rhodia has proposed two categories of amendments in case I find against them on the sufficiency argument regarding the upper limits of the claims. In the light of my findings above, these are not necessary and I shall not deal with their allowability.
- Rhodia has carried out tests on samples of Neo's C100 and C100N commercial products. It is accepted by Neo that the tests show that the physical properties of these samples fall within the numerical limits of claims 1, 3, 4 and 5, (but not 2) of the Patent.
- Rhodia relies on Neo's certificates of analysis for the samples to establish that they are "ceric oxide consisting essentially of ceric oxide". These show that ceric oxide is present as a ratio of total rare earth oxide at greater than 99.5%. Professor Burch states that:
- He therefore concludes that the samples consist essentially of ceric oxide. Neo challenge this on the basis that he has not taken account of any impurities not listed on the certificates of analysis for whatever reason. Neo also points out that the methods of analysis that would have been used cannot detect all elements, particularly carbon and fluorine. Professor Burch agreed that carbon could be present in the final product if organic surfactants had been used. His evidence is that the skilled person would normally aim to remove the carbon from the final product. Neo say that it does not follow that it is necessarily all removed in practice.
- I find that on a balance of probabilities there are no relevant additives present in Neo's products and that they infringe claims 1, 3, 4 and 5 of the Patent.
- The Patent does involve an inventive step.
- The Patent is not insufficient.
- The proposed amendments are not necessary.
- The Defendant's products infringe claims 1, 3, 4 and 5 of the Patent.
Roger Wyand QC, Deputy High Court Judge :
Technical Background
The Skilled Person
"I consider that the Skilled Person would be a graduate chemist with experience in the design, preparation and characterisation of metal oxide supported catalysts. He would have several years' professional experience (most likely in industry) in the synthesis and characterisation of catalytic materials either at a catalyst manufacturer, an oil company or a chemicals/materials manufacturer. The Skilled Person would have knowledge of chemistry, the synthesis of metal oxides and catalysts, the characterisation of materials in general and catalysts in particular and also how these materials (metal oxides and catalysts) are applied in their particular sector of industry."
"As discussed more fully below, the Patent is principally directed to the use of ceric oxide for purifying vehicle exhaust gas. Although the Patent explains (in [0001]) that the invention relates to "ceric oxide that has excellent heat resistance useful for catalysts, functional ceramics, solid electrolyte for fuel cells, and the like, and particularly suitable for use as a co-catalyst material in catalysts for purifying vehicle exhaust gas " the skilled person would understand it to be concerned principally with those applications of ceric oxide where its oxygen storage capacity and thermal stability at high temperatures are important. Rhodia contends that the skilled person would recognise that the use to which the parameters of the Patent were relevant is vehicle exhaust catalysts. Individuals not working in the field of vehicle exhausts will be less interested in the Patent and the prior art than those working in the field."
"In the case of obviousness in view of the state of the art, a key question is generally "what problem was the patentee trying to solve?" That leads one in turn to consider the art in which the problem in fact lay. It is the notional team in that art which is the relevant team making up the person skilled in the art. "
The skilled person, if he was in automotive
12 might know, but the skilled person making all the other
13 catalysts, which there are a lot of those people as well
14 interested, I do not think they would be aware or they would
15 be able to pick out a number after calcination at 900.
16 Q. Because this patent is not really directed at them?
17 A. In their use of ceria and their preparation of ceria, they
18 would never have taken it anywhere near 900.
19 Q. Yes. So this patent is not really directed then -- claim 1 of
20 this patent is to calcination at 900, and that is why this
21 patent is not really directed at those sort of ----
22 A. They would not be interested in that, no.
The Experts
The Witness of Fact
The Common General Knowledge The Sources
4.9 I would note that industrial chemists working on well-defined projects do not tend to look far beyond their own immediate areas. Therefore they frequently miss advances in other areas that later prove to be key enabling technologies in their own areas.
4.54 A person working in an area of technology that utilises catalysts will necessarily have a specific area of interest focussing on a particular set of catalytic reactions used in their area of technology.
4.55 As such, publications and other information outside a skilled person's area of interest or involving different reactions will be of little relevance to them. In particular, publications where the reaction conditions, catalyst material, and/or type of reaction being catalysed differ to those used in the skilled person's area of interest are unlikely to be considered.
4.56 For example, catalyst technologists working on automotive exhaust catalysts will have little interest in FT catalysts, and both these would be completely uninterested in olefin polymerisation catalysts. The reactions to be catalysed and the reactor types in which they must work are very different with limited overlap in interests.
The Common General Knowledge properties and applications of cerium oxide
i) Ceria is used in a number of catalytic applications (i) as a catalyst itself, (ii) to stabilise other metal oxides and dispersed metal particles against sintering, and (iii) to store and release oxygen;
ii) The use of essentially pure ceric oxides suffered from poor thermal stability and by 2001 mixed oxides and cerium zirconium mixed oxides in particular, were used as the oxygen storage co-catalyst in catalytic converters for petrol vehicles because of the high temperatures involved;
iii) Attempts had been made to improve the resistance to sintering of pure ceric oxide by altering its method of preparation or post-preparation but the addition of other oxides was known to be the most effective way to reduce sintering;
iv) Mixed oxides could have a lower surface area than pure ceric oxide at lower temperatures;
v) Other dopants such as silica had also been used to give thermal stability to high surface area oxides;
vi) The most commercially important application of ceric oxide at the priority date was as a component of exhaust catalysts in the form of a ceria/zirconia mixed oxide;
vii) Ceric oxides were also used in fluidised catalytic cracking in which SO2 is converted to H2S and in ethylbenzene dehydrogenation in the production of styrene. In these applications thermal stability above 8000 C was not particularly important.
The Common General Knowledge Metal Ion Hydrolysis and 'Free Acidity'
The Patent
"[0012] It is therefore an object of the present invention to provide ceric oxide that has excellent heat resistance and oxygen absorbing and desorbing capability useful as a co-catalyst material suitable for purifying exhaust gas, that is capable of maintaining a large specific surface area even in use in a high temperature environment, yet capable of exhibiting high oxygen absorbing and desorbing capability also in a lower temperature range, a method for preparing such ceric oxide, and a catalyst for purifying exhaust gas utilizing such ceric oxide.
[0013] .Thus the inventors, have made intensive efforts to increase the crystallinity of the precursor, and found out a reaction method for improving the crystallinity of the precursor at a high temperature under an oxidizing atmosphere, to thereby complete the invention."
4 Q. Do you agree that the characteristics of the materials claimed
5 in claims 1 to 5 would have been of considerable interest to
6 the skilled person at the priority date?
7 A. Yes.
8 Q. Because up until then the skilled person would have had no
9 idea how to make ceric oxide which retained such a high
10 surface area following calcination at high temperatures
11 without the addition of a dopant?
12 A. At that time, probably, yes.
13 Q. So, this was a significant contribution to the art?
14 A. Yes.
i) providing a cerium solution not less than 90 mol% of which cerium ions are tetravalent;
ii) holding said cerium solution prepared in step (a) at 60 to 220°C under heating;
iii) cooling said heated cerium solution;
iv) adding a precipitant to said cooled cerium solution so as to make the pH of the solution not lower than 7, to thereby obtain a precipitate; and
v) calcining said precipitate.
"Example 1
[0050] A ceric nitrate solution not less than 90 mol% cerium ions of which were tetravalent was taken so that 20g of cerium in terms of cerium oxide was contained, and the total volume was adjusted to 1 liter with pure water. Here, the concentration in terms of cerium oxide was 20 g/L. The solution was placed in an autoclave reactor, heated to 100 °C, held at this temperature for 24 hours, and allowed to cool in an atmosphere to room temperature.
[0051] Then an aqueous ammonia solution was added to neutralize to pH 8 to obtain cerium oxide hydrate in the form of a slurry. The slurry was then subjected to solid-liquid separation with a Nutsche filter, followed by separation of the mother liquor, to obtain a filter cake. The filter cake was calcined at 300 °C for 10 hours in a box-type electric furnace under air atmosphere to obtain ceric oxide, which was then ground in a mortar into ceric oxide powder (referred to as powder (A) hereinbelow). The specific surface area of powder (A) was measured by the BET method. Further, the specific surface areas of powder (A) after calcination at 800 °C for 2 hours, at 900 °C for 5 hours, and at 1000 °C for 5 hours, respectively, were measured by the BET method. The tap density and total pore volume of powder (A) were also measured. Further, powder (A) was calcined at 900 °C for 5 hours, and then the OSC of the resulting ceric oxide powder was measured at 400 °C. The results of these measurements are shown in Table 1.
[0052] Powder (A) was calcined at 1000 °C for 5 hours, and then the TPR measurement was made. The results are shown in Fig. 1. Further, from the TPR curve taken after calcination at 1000 °C for 5 hours, the ratio of the area (S1) defined by the baseline and the TPR curve in the temperature range of 200 to 600 °C to the area (S2) defined by the baseline and the TPR curve in the temperature range of 600 to 1000 °C, i.e., the S1/S2 ratio, was determined. The results are shown in Table 1."
Anticipation
Obviousness The Law
(1) (a) Identify the notional "person skilled in the art"
(1) (b) Identify the relevant common general knowledge of that person;
(2) Identify the inventive concept of the claim in question or if that cannot readily be done, construe it;
(3) Identify what, if any, differences exist between the matter cited as forming part of the "state of the art" and the inventive concept of the claim or the claim as construed;
(4) Viewed without any knowledge of the alleged invention as claimed, do those differences constitute steps which would have been obvious to the person skilled in the art or do they require any degree of invention?
Obviousness Applying the Pozzoli test
The Prior Art Relied On
"Cerium oxide and zirconium oxide are known compounds
that are particularly useful constituents, either alone or in
combination, in a wide variety of catalyst compositions, e.g.,
multifunctional catalyst compositions, especially catalysts
suited for the treatment or conversion of exhaust gases
emanating from internal combustion engines."
"Accordingly, a major object of the present invention is the provision of novel Ce02/Zr02 mixed oxides of the solid solution type, having large specific surface areas, and this over a wide range of compositions, in particular at high contents of zirconium.
Another object of the present invention is the provision of such novel Ce02/Zr02 mixed oxides which retain a large specific surface area even after calcination(s) at elevated temperatures.
Still another object of this invention is the provision of particular synthetic technique for the preparation of said novel Ce02/Zr02 mixed oxides."
i) providing a mixture, in aqueous solution, in the required stoichiometric proportions, of soluble compounds of cerium and of zirconium, and optionally of yttrium;
ii) heating the mixture thus formulated;
iii) recovering the reaction product thus obtained; and
iv) if appropriate, calcining the reaction product thus recovered.
i) Column 6, lines 18 to 22 describe the optional addition of aqueous ammonia solution to permit the recovery yields to be increased.
ii) Column 6, lines 23 to 26 describe the option of repeating the heating stage one or more times in cycles of thermal treatments. The product is recovered, optionally washed and dried in air between 80 and 350°C until a constant weight is attained.
iii) An optional calcination step is described in column 6, lines 34 to 46, with conditions depending on the subsequent application of the material, taking account of the fact that the specific surface area of the product is so much the lower because the temperature of calcination employed is higher.
"8.17 Examples 1 and 3 involve neutralisation of the initial free acidity of the cerium (IV) solution and the addition of further base according to a formula which gives "a ratio of neutralisation, r" not exceeding 0.5 In examples 2, 4 and 5 the initial free acidity is not neutralised and no further base is added at this stage.
8.18. Example 1 involves a single autoclave step at 160ºC for 4 hours followed by filtration, washing with ammonia and drying at 80ºC.
8.19. Examples 2 and 3 involve a first autoclave step at 150ºC for 4 hours followed by addition of base to pH 9.5, recovery of the product by filtration, re-suspension of the solid product in water, heating to 100ºC for 1 hour and filtering off the product.
8.20. Examples 4 and 5 involve a first heating of the solution to 100ºC for 2 and a half hours, a cooling step and addition of base to pH 9 to 9.5 followed by a second heating step at 100ºC for 1 hour, cooling and recovery of product by filtration."
"[In Hallen v Brabantia [1991] RPC 195] the prior art disclosed a type of corkscrew to which PTFE (the well-known non-stick plastic) had been applied to make it easier to penetrate the cork. It was obvious that the same advantage would apply to all corkscrews. What was not foreseeable was that there was an extra advantage for a "self-puller" type of corkscrew. But a PTFE coated self-puller was obvious nonetheless."
n1 is the number of moles of cerium added to water, n2 is the number of moles of OH- ions required to neutralise the initial free acidity contributed by the solution of salt of cerium IV and n3 is the total number of moles of OH- contributed by the addition of the base. It explains that for total precipitation of the hydroxide species Ce(OH)4, r=4. Thus n2 must be referring to the acidity coming from any additional acid and cannot be acidity coming from hydrolysis of the cerium ions.
i) That the Patent is directed to producing pure ceria with the properties previously only obtained using mixed oxides;
ii) That the one example in Chopin directed to producing pure ceria does not give the concentration of the starting solution and does not identify the relevant properties of the ceria produced;
iii) The attempt by Neo to rely on the results obtained in the examples of the Patent to establish that the method of Chopin would produce ceria with the required properties fails because the evidence fails to establish that the skilled person would have adapted the method of Chopin to fall within the parameters of the examples in the Patent;
iv) The skilled addressee in the automotive catalyst field would not have seen Chopin as teaching the possibility of obtaining improved thermal stability for ceria but only for mixed oxides;
v) The skilled addressee in the more general catalysis field would not have been interested in Chopin as it was directed to obtaining improved thermal stability for mixed oxides and, although it used the method to produce ceria without other oxides, it gave no indication that the resulting product was of any interest save as a means to identify the crystal structure of the mixed oxides of the invention.
Insufficiency Consisting Essentially of Ceric Oxide
1.2.3 As regards the wording "consisting essentially of", which is also part of claim 1, it was not contained in any of the granted claims. Therefore, the alleged lack of clarity of this term raised by Appellant II can be objected in opposition appeal proceedings.
However, as reported in the Case Law of the Boards of Appeal of the EPO, 6th edition (2010), II.B.5.2, referring to decisions T 759/91 and T 522/91, the wording "consisting essentially of", at variance with the wording "comprising substantially", has a defined meaning because of the unequivocal character of the words "consisting of"; therefore, the use of the unequivocal words "consisting of" in combination with "essentially" has been found to solve all interpretation problems of the unclear claims examined in such previous cases. The cited decisions T 759/91 (point 2.2. of the reasons) and T 522/91 (point 2.2. of the reasons) both refer to the decision T 472/88, wherein it was decided that the term "consisting essentially of" was clear and allowed the presence of other components in a claimed composition in addition to the components mandatory in the claim, provided that the essential characteristics of the claimed composition are not materially affected by their presence (see point 3 of the reasons).
The Board agrees with these previous decisions. Therefore, even though the word "essentially" does not identify precisely the amounts of additional components which could still be contained in the claimed viscoelastic fluid, the wording "consisting essentially of" allows in the present case that the composition of claim 1, which must be viscoelastic, consists of the mandatory components listed in the claims and can contain additionally only other components which do not materially affect the essential viscoelastic characteristics of the composition, e.g. minor amounts of impurities as submitted by Appellant I during oral proceedings.
"124. The claim appeared to assume that all uEPOs had effectively the same molecular weight, irrespective of source and method of isolation. This had been shown not to be the case. So which uEPO did the claim require to be used for the test? Simply to use the first uEPO which came to hand would turn the claim into a lottery. On the other hand, it would be burdensome to have to work one's way through several specimens of uEPO (which were, as I mentioned at the beginning of my speech, extremely hard to come by) and even then the result would be inconclusive because non constat that some untried specimen did not have a different molecular weight.
125. The judge decided that the lack of clarity made the specification insufficient. It did not merely throw up the possibility of doubtful cases but made it impossible to determine in any case whether the product fell within the claim. The invention was not disclosed "clearly enough and completely enough for it to be performed by a person skilled in the art": section 72(1)(c).
126. The Court of Appeal disagreed. They said that it was sufficient that some uEPO could be tested against eEPO by SDS-PAGE. The fact that it did not specify which uEPO and that choosing one uEPO would bring the product within the claim and another would not was "lack of clarity dressed up to look like insufficiency." For my part, I do not think that can be right. If the claim says that you must use an acid, and there is nothing in the specification or context to tell you which acid, and the invention will work with some acids but not with others but finding out which ones work will need extensive experiments, then that in my opinion is not merely lack of clarity; it is insufficiency. The lack of clarity does not merely create a fuzzy boundary between that which will work and that which will not. It makes it impossible to work the invention at all until one has found out what ingredient is needed."
Insufficiency Biogen Free Beer
121 I recently reviewed the authorities on this topic, and in particular the decisions of the House of Lords in Biogen Inc v Medeva Plc [1997] R.P.C. 49 ; Kirin-Amgen v Hoechst and Generics (UK) Ltd v H. Lundbeck A/S [2009] UKHL 12, [2009] RPC 13 , at length in MedImmune Ltd v Novartis Pharmaceuticals UK Ltd [2011] EWHC 1669 (Pat) at [459][484]. For convenience, I shall briefly repeat the main points of that analysis.
122 The main points which I drew from Lord Hoffmann's opinion in Biogen v Medeva were as follows:
(i) A claim will be invalid for insufficiency if the breadth of the claim exceeds the technical contribution to the art made by the invention. It follows that it is not necessarily enough to disclose one way of performing the invention in the specification.
(ii) The breadth of the claim will exceed the technical contribution if the claim covers ways of achieving the desired result which owe nothing to the patent or any principle it discloses. Two classes of this are where the patent claims results which it does not enable, such as making a wider class of products when it enables only one and discloses no principle to enable the others to be made, and where the patent claims every way of achieving a result when it enables only one way and it is possible to envisage other ways of achieving that result which make no use of the invention.
(iii) The patent in Biogen v Medeva was invalid because it was an example of the second class of objectionable claim.
123 The key point which emerges from Lord Hoffmann's opinion in Kirin-Amgen v Hoechst is his explanation at [112] of what he had meant by "a principle of general application" in Biogen v Medeva :
"In my opinion there is nothing difficult or mysterious about it. It simply means an element of the claim which is stated in general terms. Such a claim is sufficiently enabled if one can reasonably expect the invention to work with anything which falls within the general term."
124 I summarised the reasoning of the House in Generics v Lundbeck as follows:
(i) The House agreed with Lord Hoffmann in Biogen v Medeva that it was important for United Kingdom patent law to be aligned, so far as possible, with the jurisprudence of the EPO. Furthermore, the House also agreed with Lord Hoffmann that the statement of principle which he quoted from Exxon/ Fuel oils correctly stated the law.
(ii) The House considered that the instant case was to be distinguished from Biogen v Medeva because it was concerned with a claim to a single chemical compound whereas Biogen v Medeva concerned a product-by-process claim of broad scope.
(iii) It was a mistake to equate the technical contribution of the claim with its inventive concept. In the instant case, the technical contribution made by claims 1 and 3 was the product, and not the process by which it was made, even though the inventive step lay in finding a way to make the product. It followed that the breadth of the claim did not exceed the technical contribution which the invention made to the art.
"61 So, for example, if a man finds a particular way of making a new substance which is 10 times harder than diamond, he cannot just claim "a substance which is 10 times harder than diamond." He can claim his particular method and he can claim the actual new substance produced by his method, either by specifying its composition and structure or, if that cannot be done, by reference to the method (see Kirin-Amgen at [90-91]) but no more. The reason he cannot claim more is that he has not enabled more he has claimed the entire class of products which have the known desirable properties yet he has only enabled one member of that class. Such a case is to be contrasted with the present where the desirable end is indeed fully enabled that which makes it desirable forms no part of the claim limitation."
Biogen Numerical Limits
Application to Amend
Infringement
"none of the other constituents listed on the certificates of analysis are present at a level that would have a significant effect on the specific surface area of the product when measured after calcining it at 900°C for 5 hours. In particular, the amount of silicon in the Commercial Samples is less than 0.005% for C100 and less than 0.02% for C100N. These amounts of silicon are insignificant and consistent with silicon being incorporated into the Material from the original source Minerals."
Summary