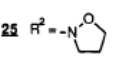Mr Justice Birss :
|
Topic
|
Paragraphs
|
|
Introduction
|
1
|
|
The witnesses
|
8
|
|
The skilled team and the common general knowledge
|
32
|
|
The lacosamide patent
|
42
|
|
Priority
|
53
|
|
Assessment – priority
|
79
|
|
The evidence
|
86
|
|
Inferences
|
107
|
|
Obviousness
|
125
|
|
Le Gall
|
126
|
|
What to do with Le Gall
|
146
|
|
Bardel
|
209
|
|
Conclusion
|
212
|
Introduction
1.
This is a patent action. It relates to EP (UK) 0 888 289. The patent covers
an anti-epileptic drug called lacosamide. The lacosamide compound is (R)
-N-benzyl-2-acetoamido-3-methoxypropionamide. Lacosamide is a successful
medicine, with annual worldwide sales projected for next year to reach €1
billion. The patent application was filed on 17th March 1997
claiming priority from a US filing on 15th March 1996. The patent
expired on 18th March 2017. There is a supplementary protection
certificate (SPC/GB09/007) which means that protection for lacosamide in this
jurisdiction continues until 2022.
2.
The claimant (Accord) is a generic pharmaceutical company. It contends
that the patent is invalid. If it is right then the SPC will be revoked,
clearing the way for generic competition. The defendant (RCT) is a technology
investment and management company based in the USA. In effect the defendant
works as a technology transfer office for a number of universities. This case
concerns work done at the University of Houston by Professor Harold Kohn and
his group. Prof Kohn and his group had been working on anticonvulsant
compounds since the 1980s. The patent came out of that work. It is
exclusively licensed to the pharmaceutical company UCB, who sell lacosamide to
treat epilepsy under the brand name VIMPAT. A share of the licence fees paid
to RCT under the licence goes back to the university and to Prof Kohn and the
other relevant workers in the group.
3.
Accord contend the patent is invalid on two grounds. Accord challenges
the patentee’s legal entitlement to claim priority from the 1996 priority
document. If that challenge is successful then a paper by Choi becomes
relevant prior art since it was published after the priority date but before
the filing date. That paper makes lacosamide available to the public and there
is no dispute that if priority is lost, the patent is invalid.
4.
Accord’s second challenge is obviousness. This is based on the state of
the art before the priority date, which included a number of papers and other
publications from Prof Kohn’s group relating to their work on anticonvulsant
compounds. Accord relies on two starting points for the obviousness analysis.
One is a master’s thesis by a student called Philipe Le Gall. It is entitled 2-Substituted-2-acetamido-N-benzylacetamides
Synthesis. Spectroscopic and Anticonvulsant Properties. It was made
available to the public in December 1987. Le Gall was a student of Prof Kohn.
The other attack is based on a paper referred to as “Bardel” the full citation
of which is given below. That also came from the work of Prof Kohn’s group.
5.
The attack based on Le Gall was originally put in two ways but by the
closing Accord had narrowed its case over Le Gall to a single approach. Accord
submits that it would be obvious for the skilled team given Le Gall at the
priority date in 1996 to do a literature search to find what had come out of
Prof Kohn’s group in the nine years since the thesis. They would find a number
of publications from the group. Accord contends that lacosamide is obvious
over Le Gall taking that supplementary information into account. The other
approach to Le Gall which had been part of Accord’s case until closing was to
rely on the thesis alone and not the supplementary information. That case was
abandoned.
6.
RCT denies the allegation of lack of entitlement to priority and denies
that the invention is obvious.
7.
There had been further points or squeezes on insufficiency and
substantive priority concerning chronic toxicity but by the closing it was
common ground they did not need to be addressed.
The witnesses
8.
Accord called two technical experts: Professor Brian Cox, a medicinal
chemist and Mr Reece Jones, a toxicologist.
9.
Professor Cox is Professor of Pharmaceutical Chemistry at the School
of Life Sciences, University of Sussex. Professor Cox received his PhD at the University of Manchester in 1986. He then had extensive experience in industry first at
Schering-Plough and then at Glaxo Research Group from 1990. Whilst at Glaxo
Research Group, Professor Cox worked on various of their medicinal chemistry
programs, and was promoted to Research Leader in 1995. One of his projects
was a study starting in September 1995 to look for analogues of the
anticonvulsant sodium-channel blocker lamotrigine, seeking a treatment with
improved tolerability. Around the same time, he was also lead chemist on a
project to identify the molecular target of a competitor anticonvulsant
product, retigabine and also worked on a project concerning calcium channel
agonists as potential anticonvulsants. His Professorship at the University of Sussex started in 2014.
10.
RCT levelled a number of criticisms of Professor Cox. The major ones
were that: his positions were at odds with the textbooks, his views were
influenced by hindsight, and he gave long answers which did not answer the
questions put. I reject all of RCT’s criticisms, for the following reasons.
Professor Cox did not accept certain points in textbooks but that was an
expression of his genuine views which he explained clearly. On hindsight, I do
not accept Accord’s case that Professor Cox’s opinions on what is obvious over
the prior art were fully formed without any reference to the patent. If it
matters (and I believe it does not) the evidence did not establish that.
However I also reject RCT’s corollary that at some overall level the fact that
his views were formed after he had read the patent means they are necessarily
tainted with hindsight. Professor Cox understood that the court’s task in
trying an obviousness case is to work out what would be obvious, without
hindsight. He sought to help the court by explaining his opinions about that.
As for long answers not answering the question, Professor Cox did give quite
long answers but that was borne of his desire to ensure that the court
understood his full opinions on the topic. Also it was not helped by occasions
on which a question was put by presenting a summary of what the Professor’s
report was said to say but which missed out relevant details.
11.
Professor Cox was a good witness, seeking to help the court resolve the
issues in this case.
12.
Mr Reece-Jones is a consultant toxicologist who trained as a
toxicologist following receiving his HND in Applied Biology from Manchester
Polytechnic. He started training at Hazleton in 1979, and he has worked both
in-house in industry and at consulting toxicology companies. He worked as a study
director since 1988. He has worked on a wide variety of compounds antibiotics,
antihistamines, and central nervous system (CNS) compounds. He is a highly
experienced toxicologist and was well placed to give evidence as to toxicology
in 1996.
13.
Mr Reece-Jones was barely cross-examined. By closing the point to which
his evidence went had been dropped. I thank him for his attendance at court.
14.
RCT called two technical experts: Professor Wolfgang Löscher, a
pharmacologist and Professor Simon Ward, a medicinal chemist.
15.
Professor Löscher is Professor and Director of Pharmacology, Toxicology
and Pharmacy at the University of Veterinary Medicine, Hannover. He is also the
founder of the Centre of Systemic Neurosciences there. He has worked in the
field of anticonvulsant drugs since the 1970s. He has worked primarily as an academic
but his work has always involved close links with industry and drug
development.
16.
Accord submitted that Professor Löscher’s experience with drug
development was primarily as a consultant brought in only when the drugs were
already at a stage of pre-clinical development or later. The contrast was
between that and experience of an earlier stage of drug development. Part of
Professor Löscher’s extensive work in this field did involve the tasks referred
to by Accord, but I reject the submission that this qualifies his expertise.
He knew very well how the in vivo pharmacological assays such as the MES
test (see below) were used in early stage drug development and was well
qualified to express opinions about all the issues he covered in this case.
17.
Accord submitted he was an ardent advocate of criticism of the MES test
in particular especially for its tendency, as he saw it, to produce “me too”
drugs. So he was. I was not satisfied that this view of the MES test
represented the common general knowledge at the relevant time. He also had
strong views about the need to address drug resistant epilepsy in particular as
opposed to treating epilepsy in general and again I am not persuaded those
views represented the common general knowledge of a drug development team in
1996. He also had worked with and visited the NIH and had personal knowledge
about the results of a comprehensive programme of testing putative
anticonvulsant compounds for drug developers which the NIH had been running for
a number of years by 1996. I will deal with that in context if need be.
18.
Professor Löscher was an excellent witness, seeking to help the court at
all times.
19.
Professor Ward is Visiting Professor of Medicinal Chemistry in the
School of Life Sciences at the University of Sussex and is also Sêr Cymru
Professor in Translational Drug Discovery and Director of Medicines Discovery
Institute at Cardiff University. After graduating in chemistry in 1993
Professor Ward worked at the pharmaceutical company Chiroscience for a year and
then went back to Cambridge to do his PhD (1994-1997). He then worked at the
pharmaceutical company Cerebrus until moving to Knoll in 1999 and on to GSK in
2001 where he remained until taking up his Professorship at the University of
Sussex.
20.
Professor Ward was another good witness, seeking to help the court.
21.
At the priority date (1996) Professor Ward was studying for his PhD.
Accord submitted he was not in a position to assist the court about the
attitudes, interests and common general knowledge of those in the field at the
relevant date. Accord submitted his evidence about this could not be relied
on. I do not accept the full extent of that submission. I distinguish between
two different things. In my judgment Professor Ward was able and was well
qualified to assist the court in relation to medicinal chemistry itself, in
other words the way medicinal chemists apply their chemical knowledge and skill
to the problems presented by pharmaceutical drug development. He is an expert
medicinal chemist and was close enough to the field at the time to be able to
assist the court. He was good at explaining things. Medicinal chemistry as a
discipline did not change over the relevant period in a manner which would
undermine Professor Ward’s ability to assist. The fact he did not work on
anticonvulsant or CNS drugs until much later does not matter. Medicinal
chemists would routinely be called upon to work in a new field. Despite hints
to the contrary by Accord, I find that Professor Ward’s work since he graduated
in 1993 has all been in or closely related to medicinal chemistry, with some
synthetic chemistry as many medicinal chemists would also undertake
particularly at the start of their careers.
22.
However where the nature and timing of Professor Ward’s personal
experience is relevant and does qualify his ability to help is in addressing
evidence about the general approaches of drug development teams in
anticonvulsant medicine in 1996. He was not there.
23.
The parties also called experts on US federal and state law. They were
not cross-examined.
24.
Accord relied on Professor Robert Merges for US federal law; and Judge
Leonard Davis for the law of the state of Texas.
25.
Professor Merges is the Wilson Sonsini Goodrich & Rosati Professor
of Law and Technology at US Berkeley School of Law, a position he has held
since 1995. He teaches Intellectual Property, Patent Law and Contracts and his
research focusses on the economic aspects of intellectual property rights, with
a focus on patents.
26.
Judge Davis was the Chief Judge of the United States District Court for
the Eastern District of Texas. That court is famous for its experience of
patent law. From 2002-2015 Judge Davis managed one of the busiest dockets in
the United States handling over 1700 patent cases a year, and personally tried as
many as eleven jury trials in one year. The judge was also the Chief Judge of
the Twelfth Court of Appeals of the State of Texas. He has now retired from
the bench and works as Of Counsel at the law firm Fish & Richardson.
27.
RCT’s US law experts are: Professor Donald Chisum on US federal law and Judge
David Folsom on Texas state law.
28.
Professor Chisum has held various professorships at Stanford University,
the University of Washington and Santa Clara University, each of which being
Professor of Law. He is the author of Chisum on Patents, the well known text
book on US patent law.
29.
Judge Folsom is currently a partner in the law firm Jackson Walker LLP.
From 1995 till 2012 he served as a United States District Court Judge, Eastern
District of Texas (as Chief Judge from 2009-2012). During this time he heard
over 250 trials and has presided over hundreds of patent cases.
30.
Rightly, neither side suggested the US law experts were not qualified to
give the evidence they did.
31.
RCT also called a number of fact witnesses, who were not
cross-examined. Their evidence related to the entitlement to priority issue.
The witnesses were Professor Kohn himself, Dr Ramanan Krishnamoorti of the University
of Houston and Mr Timothy Reckart formerly of RCT.
The skilled team
and the common general knowledge
32.
In this case the person skilled in the art is a team. The team will
include a medicinal chemist and a pharmacologist as well as other disciplines.
33.
The only aspect of chemistry worth mentioning at this stage is
chirality. Many of the compounds in this case are chiral. Chiral compounds
exist in two non-superimposable mirror image forms known as enantiomers. The
compounds have a carbon atom with four bonds in a tetrahedral shape and
different substituents attached to each of the four bonds. Chirality is common
in biological systems and in many cases one enantiomer may have a completely
different effect from another enantiomer. A racemate is an equal mixture of
the two enantiomers.
34.
Turning to clinical issues, the skilled team would know that epilepsies
can be categorised by reference to the types of seizure that the patient
suffers from. These can be broadly classed as partial or generalised seizures.
The former arise in a localised area of the brain and are further classified
as: “simple” (where consciousness is maintained); “complex” (where
consciousness is lost or impaired); and “secondarily generalised” (involving
convulsions). Generalised seizures arise in all or large parts of both
cerebral hemispheres and comprise two main categories: “tonic-clonic”/“grand
mal” (involving widespread convulsive activity); and “absence”/”petit mal”
(where consciousness is impaired with little or no motor disturbance).
35.
The treatment of epilepsy involves the administration of an
anticonvulsant drug or cocktail of drugs. These are given chronically, with a
view to preventing or reducing the incidence of seizures.
36.
Various anticonvulsant drugs had been identified by the Priority Date
including the “first generation” anticonvulsants phenytoin, phenobarbital and
ethosuximide which had been discovered in the 1930s/1940s using animal models.
Phenytoin remained a mainstay of the treatment of epilepsy at the priority
date. Various other drugs were discovered in the “second generation” of
anti-epileptic drugs, in the 1950’s and 60’s, including carbamazepine, sodium
valproate and benzodiazepines. These too were found using animal models.
37.
In the early 1990s, a number of new anticonvulsant drugs came on the
market including vigabatrin, gabapentin, lamotrigine, clobazam, topiramate,
felbamate, tiagabine, oxcarbazepine, zonisamide and progabide. Nevertheless,
for the skilled team there remained a need for new and better anticonvulsants
at the priority date.
38.
To a pharmacologist interested in anticonvulsant drugs, there were
various assays based on animal models which could be performed to evaluate the
possible activity of candidate compounds. One was the MES test. MES stands
for Maximum ElectroShock. It was a test in mice or rats in which a seizure is
induced using a standardised electric shock, and the ability of a compound to
prevent the seizure is measured. The effect of the drug is measured at a time
after delivery called TPE (time of peak effect). The seizure is determined by
HLE (hind limb extension) in the test animal. For a compound which is
effective, the MES test produces an ED50 value. That is the dose in mg/kg
require to protect 50% of the animals challenged. So a low ED50 is more
desirable as it represents a higher potency.
39.
Other kinds of assay used involved a chemically induced seizure instead
of a seizure induced by an electric shock. An example is the sc Met test. The
term sc Met refers to the subcutaneous (sc) administration of the compound
Metrazol (pentylenetetrazol). That compound induces the seizures. The test is
not the same as the MES test but it still produces an ED50 value. Other
similar tests using a chemically induced seizure are the sc Bic and sc Pic
tests. These use different compounds to induce seizures.
40.
A different kind of animal test which was also routine was to see if the
compound caused undesirable neurological symptoms. Put simply the point is
that one may be able to treat seizures by giving a drug which acts as a
sedative to the patient; however what is wanted is a drug which can treat the
seizures without a major sedative effect. The possible sedative effect is
assessed using these tests. One of them was the rotarod test, in which rodents
which are trained to balance upon a rotating rod, are given the drug, and the
unwanted neurotoxicity assessed by monitoring the extent to which the animal
can stay on the rod. The other major test used was the horizontal screen test,
in which the animals are placed upon a wire mesh that is slowly rotated 180o.
If the animals are unable to climb to the top of the screen in a certain period
of time, this also demonstrates neurological impairment. Using these kinds of
neurotoxicity tests it is possible to calculate the toxic dose which causes
neurological impairment. This is calculated as a TD50 value.
41.
The ratio of a compound’s ED50 value to its TD50 value is known as its
protective index (PI). The skilled team would want a compound with a good
protective index, to reduce the neurological toxicity issues with the compound
– this was a key selection criterion.
42.
The skilled team looking for promising new anticonvulsant compounds
would gauge the putative compound’s test results (ED50, TD50 and PI) in animal
models such as the MES test against the results for known compounds, such as
phenytoin. If the results were promising the team would screen good compounds
in further animal models such as sc Met. Over time the ED50 levels in the MES
test which a team would consider worth pursuing were dropping. Recognising
that it is an over simplification to pick a value and state that the team would
focus on that, nevertheless by 1996 a fair picture is presented by considering
a team looking for ED50 values in the MES test of around 10 mg/kg or less along
with a good PI.
The lacosamide
patent
43.
The patent starts with a statement that the invention relates to novel
enantiomeric compounds and pharmaceutical compositions useful for treating
epilepsy and other CNS disorders. The background section describes the use of
anticonvulsant drugs to treat seizures, and summarises the two main kinds of
seizures. Various existing drugs such as phenytoin and phenacemide are
mentioned and there is a reference to a prior patent from the Kohn group. At
paragraph [0006] the patent refers to problems with the existing agents
including poor management of the disease and disturbing side effects. A
particular point is made about the difficulty that these agents are
administered chronically and this is associated with liver toxicity. In
paragraph [0007] four criteria for an ideal anticonvulsant drug are set out:
“(1) has a high anticonvulsant activity, (expressed as a low
ED50);
(2) has minimal neurological toxicity, (as expressed by the
median toxic dose (TD50)), relative to its
potency;
(3) has a maximum protective index (sometimes known as
selectivity or margin of safety), which measures the relationship between the
doses of a drug required to produce undesired and desired effects, and is
measured as the ratio between the median toxic dose and the median effective
dose (TD50/ED50);
and
(4) is relatively safe as measured by the median lethal dose
(LD50) relative to its potency and is
non-toxic to the animal that is being treated, e.g., it exhibits minimal
adverse effects on the remainder of the treated animal, its organs, blood, its
bodily functions, etc. even at high concentrations, especially during long term
chronic administration of the drug. Thus, for example, it exhibits minimal,
i.e., little or no liver toxicity.”
44.
The paragraph continues by explaining why, although not so critical for
short term administration, the fourth criterion is extremely important for an
anticonvulsant which is to be taken over a long period of time or in high
dosage as follows:
“It may be the most important factor in determining which
anti-convulsant to administer to a patient, especially if chronic dosing is
required. Thus, an anti-convulsant agent which has a high anti-convulsant
activity, has minimal neurological toxicity and maximal P.I. (protective index)
may unfortunately exhibit such toxicites which appear upon repeated high levels
of administration. In such an event, acute dosing of the drug may be
considered, but it would not be used in a treatment regime which requires
chronic administration of the anti-convulsant. In fact, if an anti-convulsant
is required for repeated dosing in a long term treatment regime, a physician
may prescribe an anti-convulsant that may have weaker activity relative to a
second anti-convulsant, if it exhibits relatively low toxicity to the animal.
An anti-convulsant agent which meets all four criteria is very rare.”
45.
Next is a summary of the invention section which corresponds to claim
1. Although the patent is drafted on the basis of a class of compounds defined
in claim 1, the extent of the class in claim 1 does not matter because claims 8
to 10 relate to lacosamide specifically. The remainder of the summary of the
invention section states that administering the compounds of the invention
provide an excellent regime for the treatment of epilepsy and some other CNS
disorders.
46.
The detailed description section starts with the general synthesis of
the compounds. The synthesis described at Scheme 1 starts from the amino acid
D serine and preserves the chirality of the starting material. A racemic
serine can be used instead and the final product resolved to produce the
relevant R isomer (paragraph [0030]). Wide general statements are made about
dosing, route of administration, dosage forms and excipients (paragraphs [0031]
– [0042]).
47.
The examples start at paragraph [0044]. Examples 1, 2 and 5 provide
alternative syntheses for lacosamide. Examples 3 and 4 relate to other
compounds in the class of claim 1 (they are fluorinated). Comparative examples
1 – 13 relate to other similar compounds which are not within the claimed
class.
48.
Pharmacology is dealt with from paragraph [0077] and the results of
various animal studies are in Table 1. They show testing of lacosamide and two
other compounds within the claimed class along with a number of comparative
example compounds. The tests are in mice (ip) and rats (po). The tests are
MES and neurological toxicity via the rotarod test. The results report ED50
and TD50 values with some 95% confidence intervals. PI values are
stated. The values for lacosamide are:
|
Compound
|
mouse (ip)
|
Rat (po)
|
|
MES
ED50
|
Tox
TD50
|
PI
|
MES
ED50
|
Tox
TD50
|
PI
|
|
Lacosamide
|
4.5
(3.7-5.5)
|
26.8
(25.5-28.0)
|
6.0
|
3.9
(2.6-6.2)
|
>500
|
>128.2
|
49.
The patent summarises the data in Table I as a whole from paragraph
[0079]-[0085] as showing that the R enantiomers of the invention have quite
potent anticonvulsant activity and an excellent drug profile while the
comparative compounds tested (save for the furyl derivative) are significantly
inferior drugs. Even if the level of neurotoxicity is low and the PI is
satisfactory, the patent explains that one wants to administer as little drug
as possible and so a low potency value is advantageous (paragraph [0085]).
50.
Turning to chronic liver toxicity, in paragraphs [0086]-[0087] the
patent explains that while both the furyl derivative and the compounds of the
invention have a good drug profile, the furyl compound is more toxic to the
animal, making it less suitable for chronic administration. The compound
called BAMP (which is lacosamide) exhibited little if any toxicity to the
animal. The animal toxicity tests on lacosamide are reported in [0089] to
[0105]. There is a short term, 48 hour, study in rats in which minimal liver
toxicity was shown. Then a longer term, 30 day, study in rats was run, which
showed no histological evidence of an adverse effect at the highest doses.
Toxicity studies are run on other compounds outside the invention. The results
for those compounds and for BAMP/lacosamide are summarised in Table 6. The
comparison favours BAMP/lacosamide.
51.
The description ends at paragraph [0141] with a statement that the
compounds of the invention have an excellent drug profile, meet all the four characteristics
outlined before and can be used for chronic administration.
52.
Claim 8 is a claim to lacosamide itself, in other words (R) -N-Benzyl
2-Acetoamido-3-methoxypropionamide. Claim 9 is to the “substantially
enantiopure” compound of claim 8. Claim 10, as dependent on claim 9, is to the
therapeutic composition of an anticonvulsant effective amount of substantially
enantiopure lacosamide and a pharmaceutically acceptable carrier.
Priority
53.
The right to claim priority is in section 5 of the Patents Act 1977.
That section is one of the sections to which section 130(7) applies and so it
is intended to have the same effect as the relevant parts of the EPC. In the
EPC priority is governed by Art 87. This in turn gives effect to Article 8 of
the PCT which in turn gives effect to Article 4(A)(1) of the Paris Convention.
The Paris Convention is the origin of the international priority right.
54.
Before getting into the law any further it is convenient to state the
problem which arises in this case. The underlying facts are not in dispute.
55.
For lacosamide the inventor was Prof Kohn. The US filing from which
priority was claimed was made by Prof Kohn on 15th March 1996. The
international application under the Patent Cooperation Treaty (PCT) filing
number PCT/US97/04579, which matured into the patent in suit, was filed by RCT
on 17th March 1997 claiming priority from that earlier filing.
There is on the face of it a properly executed written assignment of the
invention from Prof Kohn in favour of RCT dated 4th February 1997.
The assignment expressly includes the right to claim priority at clause 5. In
the context of priority it is convenient to refer to the relevant invention as
lacosamide rather than refer to the whole subject matter of the patent in suit
but nothing turns on that difference.
56.
So is everything in order? Accord say it is not. Accord took a wide
range of points prior to the trial but by the opening its case came down to a
single point. Accord contends that the assignment of 4th February
1997 only took effect as an assignment of the bare legal title to the invention
and priority claim. What it did not do was assign the equitable or beneficial
title to that property to RCT. That equitable or beneficial title was with the
University of Houston (“Houston”). It may (or may not) have ended up with RCT
later but that does not matter. Accord submits that as a matter of law, what
is required for the priority claim to be accepted in this court at this trial
is that the substantive right to priority, also known as the equitable or
beneficial title to that right, was with the correct party on the correct day.
The correct party was RCT and the correct day was 17th March 1997
when the international application was filed. Accord contends that this did
not happen and so the claim to priority should fail.
57.
The reason the substantive right to priority might have been in the
wrong place at the wrong time (if it was) is as follows. The first step is
that for inventions made by faculty members or other university staff such as
associates and employees at Houston, those individuals are obliged as a result
of the contractual arrangements between them and the university to assign them
to Houston (or to a person appointed by Houston such as RCT). That applies to
all the relevant individuals here such as Prof Kohn and the others. This is
common ground.
58.
The second step is that Houston had a long standing agreement with RCT
relating to inventions and patents. It started as an agreement in 1966 between
Houston and RCT’s predecessor. This is also common ground.
59.
By that agreement Houston had the right but not the obligation to offer
inventions to RCT. For any relevant invention made by its faculty/staff,
Houston would make a decision whether or not to offer that invention to RCT.
As between Houston and RCT it did not matter what internal Houston process was
used to make that decision. What matters is that at all relevant times under
the contract between Houston and RCT, Houston was not obliged to offer
inventions to RCT. From RCT’s point of view it was a decision in Houston’s
discretion. Conversely once Houston had disclosed and offered an invention to
RCT, RCT was not obliged to accept it, RCT could choose to accept it or not.
Again how the party (now RCT) made the decision was up to that party (RCT). If
RCT decided to accept the invention it would notify Houston of that
acceptance.
60.
From now on pursuant to the contract, RCT would file and prosecute
patent applications to protect the invention in its own name and Houston would
ensure it could do so and execute or have executed all necessary assignments.
A share of any revenue would be passed back to Houston. In fact under the
arrangement some payments of modest sums such as $3,000 at one early stage
would be made by RCT to Houston. Note that the disclosure and offering were
all confidential so none of this makes anything available to the public and
references to disclosure in this context have to be understood that way.
61.
There was clear evidence of this arrangement working as described for
some work by the Kohn group prior to lacosamide. The project was called “FAA”
which means Functionalised Amino Acid. Compounds would be identified and
tested by Prof Kohn and his group. For this purpose a group of related
compounds is an invention. Houston would consider whether to offer that
invention to RCT. In fact it did decide to do so on a number of occasions.
RCT would take the invention disclosure documents and consider them in order to
decide whether to accept them. RCT in fact did decide to accept them and file
patent applications. The compounds developed by Prof Kohn (not lacosamide)
were licensed to Eli Lilly by RCT as licensor under these patent applications.
The Lilly licence had come to an end before lacosamide.
62.
The problem, says Accord, is that there is no evidence the proper
process took place for lacosamide. There is no evidence of a disclosure by
Prof Kohn to Houston of this invention in order for Houston to make its
choice. Nor is there evidence of Houston, rather than Prof Kohn, offering the
invention to RCT having decided to do so. So, says Accord, what seems to have
happened is the following.
63.
Prof Kohn made the invention. At that stage Prof Kohn was obliged by
his obligations to the university to assign it to Houston or to their order.
So analytically (says Accord) while Prof Kohn held the legal title, Houston
held the equitable title since they had a legally enforceable right to call for
an assignment of the legal title. Accord says this analysis is correct under
US law (Federal and Texas state law). In fact Prof Kohn disclosed the
invention to RCT and RCT did apply for patents. Prof Kohn also did execute the
assignment mentioned already before the international PCT application was
filed. But, says Accord, at the point in time before Prof Kohn signed the
assignment to RCT, the equitable title was with Houston. The written
assignment was not effective to move the equitable title from Houston to RCT
and so the priority claim fails.
64.
Accord’s case rests on two points: first the legal requirements to
establish a priority right and second the factual question of what actually
happened in Texas in 1996/97 and what are the legal consequences in terms of
legal and equitable ownership of property.
65.
In order to address the first point it is necessary to understand where
English law on entitlement to priority has got to. In Edwards
Lifesciences v Cook Biotech [2009] EWHC 1304 (Pat) Kitchin J held
as follows:
“In my judgment the effect of Article 4 of the Paris
Convention and section 5 of the Act is clear. A person who files a patent
application for an invention is afforded the privilege of claiming priority
only if he himself filed the earlier application from which priority is claimed
or if he is the successor in title to the person who filed that earlier
application. If he is neither the person who filed the earlier application nor
his successor in title then he is denied the privilege. Moreover, his
position is not improved if he subsequently acquires title to the invention. It
remains the case that he was not entitled to the privilege when he filed the
later application and made his claim. Any other interpretation would introduce
uncertainty and the risk of unfairness to third parties.”
(my emphasis)
66.
In the large majority of cases, including this one, a patent applicant
starts the process of obtaining a patent by filing a patent application in
their home state (e.g. here the USA). Then a year later an international
application is filed under the PCT designating all states and claiming priority
from that earlier regular national filing. The real importance of this passage
from Edwards v Cook is in the sentence emphasised. In the
normal case described its effect is that the applicant’s title to the priority
claim must be in place by the time the international application is filed. That
is because it is that application which makes the priority claim, claiming as
the priority date the filing date of the earlier application. If the
applicant’s entitlement to priority has not been secured by that time then the
position cannot be fixed after the event. This is a critical aspect of
Accord’s case. The fact that the university and RCT today are making common
cause does not help. Perhaps (and this is pure speculation) that common cause
is the result of some accommodation reached after 17th March 1997
and therefore too late to save the priority claim.
67.
Edwards v Cook has been followed and applied at
first instance on a number of occasions including at least the following:
Arnold J in KCI Licensing v Smith & Nephew [2010] EWHC 1487 (Pat) and Idenix v Gilead [2014] EWHC 3916 (Pat),
Henry Carr J in Fujifilm v Abbvie [2017] EWHC 395 (Pat);
and me in HTC v Gemalto [2013] EWHC 1876 (Pat). In KCI
Licensing v Smith & Nephew and HTC v Gemalto
the judges (Arnold J and myself respectively) accepted a significant softening
to what otherwise might have been the rigour of the rule that the title must be
secured by the time the international application is made, by accepting an
analysis based on common law principles distinguishing the equitable and legal
title to property. If the relevant local law meant that the equitable or
beneficial title to the priority right was in the hands of the person making
the priority claim in the international application, that was held to be good
enough even though that person did not then hold the legal title under the
local law and could only perfect their title after the event.
68.
The critical passage in KCI is as follows. Arnold
J had held that on its true construction the relevant agreement there did
convey the legal title to the applicant but he went on to hold that even if
that was wrong, the agreement was effective to transfer the entire beneficial
interest. The applicant had an enforceable legal right to call for a
conveyance of the bare legal title and that made the applicant the “successor
in title” for the purposes of a claim to priority under Article 87(1) of the
EPC and Article 4(A)(1) of the Paris Convention even if KC Inc had not acquired
the bare legal title at the relevant date. After referring to a decision of
the EPO Case J19/87 Burr-Brown /Assignment [1988] EPOR
350, Arnold J held:
“71. To my mind, this makes sense. Article
4(A) of the Paris Convention and Article 87(1) of the EPC are provisions in
international treaties whose operation cannot depend upon the distinction drawn
by English law, but not most other laws, between legal and equitable title. When
determining whether a person is a "successor in title" for the
purposes of the provisions, it must be the substantive rights of that person,
and not his compliance with legal formalities, that matter.”
(Accord’s emphasis)
69.
In HTC I referred to Edwards v Cook
and noted in paragraph 132 that no-one in argument before me had challenged the
proposition that a later acquisition of title to the invention
was not enough. I referred to the above passages from Arnold J’s judgment
in KCI and stated at paragraph 134:
“134. Mr Mellor submitted that this [Arnold
J’s reasoning in KCI] showed that as long as an applicant had, at the
relevant date, what English law would characterise as a beneficial title to the
invention, even if the bare legal title had not been acquired, then the
applicant was a successor in title in the relevant sense. I did not understand
Mr Tappin to dispute that and I think he was right not to. In my judgment if
the relevant person has acquired the entire beneficial interest in the
invention at the relevant time then that should be enough to satisfy the law.”
70.
When Idenix reached the Court of Appeal Kitchin LJ
did not have to express a concluded view on the subject but expressed a
provisional view that KCI and HTC
were correct. Floyd LJ and Patten LJ agreed. Fujifilm
came after Idenix in the Court of Appeal and Henry Carr J
took the same approach.
71.
To return to Idenix, the reasoning of Kitchin LJ
was the following. He noted that Arnold J had held that equitable title was
sufficient, referring to his own KCI judgment and to mine
in HTC. Kitchin LJ then noted that a critical part of the
reasoning was the EPO decision in Burr Brown and then said
the following:
“265. Mr Acland
submits as follows. The judge's analysis starts correctly but jumps to the
wrong conclusion. The signatories to the Paris Convention have a diversity of
legal traditions and it is only the common law that distinguishes between
equitable and legal ownership. Accordingly, the treatment of equitable
interests in English law cannot have any bearing on what the signatories to the
Paris Convention meant by the expression 'successor in title' in Article 4(A).
Instead, one must search for and identify a notion of ownership and transfer of
ownership that is common to all of those signatories.
266. It is my provisional view that the
decisions on this issue in KCI and HTC are correct, that the
Paris Convention does not purport to identify the requirements for the
effective transfer of title to an invention and that these matters are left to
the relevant national law. Indeed this appears to be the approach of the Boards
of Appeal of the EPO: see, for example, T 0205/14 of 18 June 2015. In these
circumstances the notion that it is the transfer of the substantive right and
title to the invention which is important makes eminently good sense.
Nevertheless, it emerged during argument that there may be other materials and
decisions which bear on this issue and to which our attention has not been
drawn. Accordingly and having regard also to the fact that it is not necessary
in this appeal to express a final view on this issue, it seems to me that it is
better left to be decided in another case.”
72.
Although obviously personalities do not come into it, one can see a
personal tinge to Mr Acland’s submissions in this case. In Idenix
his argument which failed is effectively the converse of his argument before
me. Before me his case is that the outcome of applying local law to the facts
is that the substantive right and title to the invention were with Houston on
the day Prof Kohn signed the assignment. So while I do not believe it is
disputed that RCT has those rights today, there is no evidence RCT had them
when it made the application. If, instead of RCT, Houston had filed the
application on 17th March 1997, the assignment assigning the
invention to RCT before the filing date would not have mattered, applying this
line of cases, because even though the bare legal title was with RCT, the
substantive right remained with Houston. Houston could have then filed an
application making a valid priority claim. All Accord’s case amounts to is the
logical consequence of the decisions above.
73.
RCT’s response to this submission has two aspects. First is on the
facts. RCT submits that the court should hold that in fact the assignment by
Prof Kohn was effective to ensure that RCT acquired the substantive priority
right. This factual argument is put on two bases. One that as a matter of
fact I should infer that all the relevant steps did take place and the state of
the evidence is explained by the 20 year time gap. I am invited to hold that
Houston did decide to offer lacosamide to RCT before the assignment and RCT
accepted it and Houston directed Prof Kohn to transfer it to RCT such that the
assignment was therefore effective to assign both legal and equitable title.
The other approach on the facts is to argue that in any event RCT is equity’s
darling, the bona fide purchaser for value without notice (which under the
relevant US law is provided for by statute) and therefore the assignment was
effective to leave RCT with full title despite Houston’s equitable rights.
74.
Second RCT submitted that even if either of those points were not right,
there was nevertheless a transfer of the equitable title to RCT. However its
case on this was incoherent. RCT did submit that the proposition that a person
with full equitable title but no legal title can claim priority is not
“reversible” and does not imply that the person with legal title may not claim
priority unless they prove they also hold the full equitable interest, but
there was no analysis to back this up. There was a suggestion by RCT that in KCI
it was held by Arnold J at paragraph 68 that the legal title was sufficient.
That is not accurate. By “sufficient” RCT must mean to suggest that the judge
had held that legal title was sufficient even if the equitable title was elsewhere.
That is not what Arnold decided. In KCI the relevant
company held the equitable title anyway. RCT’s submissions on principles also
contained references to a number of equitable maxims such as “equity does not
assist a volunteer” but the submission did not make sense and the point was not
pursued. I will approach this aspect of the case as a submission that under US
law (Federal and Texas law) an implied in fact agreement to assign the
equitable interest to RCT should be found to exist and was effective to assign
the interest at the same time as the February 1997 assignment. The US law
aspects of this submission are addressed below.
75.
I find that the legal principles applicable to priority entitlement are
settled at this first instance level. They are:
i)
Usually the right to claim priority goes with the right to the
invention. That is uncontroversial.
ii)
The right to claim priority must be with the person making the patent
application in which that right is claimed when they make that claim, i.e. when
the application is filed. A later acquisition of that right cannot make good a
lack of it on the relevant date. If the right was not in place at the time
then the right is lost for all time. That is Edwards v Cook.
iii)
But if the local law applicable to rights of the applicant and the
patent application at the place and time when it was made allows for a
splitting of property rights into legal and equitable interests, then it will
be sufficient to establish an entitlement to priority if the applicant holds
the entire equitable interest at the relevant date. That is KCI,
HTC and FujiFilm and was held in the
Court of Appeal in Idenix provisionally to be correct.
iv)
A person with a legally enforceable right to call for the assignment of
the legal title to a piece of property such as an invention (or a right to
claim priority) has the equitable title to that property. When the cases refer
to the applicant holding the substantive right and title to the invention, they
are referring to this legal/equitable distinction.
76.
In my judgment Accord is right in law that following from those
principles, a person who at the relevant time and under the relevant applicable
law, acquired only the bare legal title to an invention and not the equitable
title, when the equitable title is held by another, does not then hold the
substantive right and title to the claim to priority.
77.
However I cannot help but observe that if priority is lost this patent
would be revoked over a publication by the inventor in the period between the
priority date and the filing date which I infer was assumed to be a safe thing
to do because it was assumed by everyone involved that priority would be
successfully claimed. There will be many cases like this. There is no obvious
public interest in striking down patents on this ground, unlike all the other
grounds of invalidity. The difficulty starts with the point that the title
cannot be fixed retrospectively. If I may say so the reasons given by Kitchin
J in Edwards v Cook are compelling reasons why that should
be so. However the legal/equitable analysis chips away at that principle since
what is happening in those cases is that the equitable owner’s imperfect title
on the relevant date is only perfected after the event. No doubt that is why,
in the Court of Appeal, Kitchin LJ declined to get into the issue any further
since he did not have to.
78.
I offer the following tentative suggestions. One approach could be that
the effect and devolution of the priority right has to be purely governed by a sui
generis law applicable to priority rights in all signatory states to the
Paris Convention equally and applicable in all those states regardless of
whether those states recognise a legal/equitable distinction. Flaws in the title
cannot be fixed retrospectively. That is one way of interpreting Edwards
v Cook and there are good reasons for it. However it does not sit
happily with the equitable/legal distinctions made in the later cases. An
alternative could be to apply the same approach and the same applicable law to
the priority claim as applies to ownership of the invention and the right to
the patent. In a case in which there is some doubt about the claimant’s title
to the patent itself, that title has to be perfected by the judgment e.g. by
assignment or the legal owner must be joined to the proceedings (see e.g. Baxter
v NPB [1998] RPC 250). The moment the title to the patent matters
is judgment. In this case the moment the priority claim matters could be said
to be the judgment. As far as the applicable law is concerned, under English
private international law, the law applicable to the devolution of the rights
to the invention in Texas in 1996/97 is US law, which is in fact a mix of
Federal and Texan state law. Nevertheless regardless of these tentative
suggestions, I will apply the law as it is settled at this level.
Assessment –
priority
79.
It was common ground between the US law experts that there existed
between Prof Kohn and Houston an agreement whereby the professor would assign
his rights in any relevant inventions to Houston or its designee. The parties
helpfully prepared a set of agreed principles of US law. Agreed Principle 14
was that:
“Where there is a valid and enforceable promise to assign in
the future all rights in a future invention, patent application or patent, the
promise holds equitable title in the invention, patent application or patent.
Upon the invention being made, the patent application being file or the patent
being issued the promisee is entitled to demand the transfer of the legal title
and to compel the same by way of civil proceedings. Where a promisee holds
such an equitable interest, to gain legal title to an invention, patent
application or patent the promisor must transfer legal title by a written
assignment from the promisor-assignor after the invention is made, the patent
application is filed or the patent is issued.”
80.
Applying that principle leads to the conclusion that when Prof Kohn made
the lacosamide invention, Houston held the equitable interest in it.
81.
The only dispute about US law was or seems to be about whether the
transfer of equitable title to a patent requires a “clear and unmistakable act
of assignment” in order to part with the rights. In his first report Prof Merges
refers in both paragraphs 40 and 58 to the need for a clear and unmistakable
act of assignment to transfer an equitable interest. Prof Chisum did not
agree, expressing the view in his second report (paragraph 21) that such a
requirement was inconsistent with the principle that an “implied-in-fact”
contract could convey an equitable title, which principle had been expressed by
Judge Davis and which Prof Chisum agreed with. Accord submitted there was no
inconsistency between the view that an “implied-in-fact” contract could convey
an equitable interest and Prof Merges’ opinion, because the implied-in-fact
contract was simply an example of the necessary clear and unmistakable intent
required by Prof Merges being found to exist evinced by conduct.
82.
RCT relied on findings of US law in the judgment of Henry Carr J in Fujifilm
under s4(2) of the Civil Evidence Act 1972. The point is that although foreign
law is a question of fact, under s4(2)(b) the foreign law can be taken in this
trial to be in accordance with the earlier judgment unless the contrary is
proved. The two points decided by Henry Carr J as US law were:
i)
That there are no special requirements under US law for: (i) a person or
company to be nominated under an agreement to be the beneficiary of rights; or
(ii) that party to be expressly identified in a contract providing for
assignment to a nominee before the agreement can take effect (FujiFilm
paragraph 49); and
ii)
implied-in-fact contracts are not limited to the field of employed to
invent (FujiFilm paragraph 55).
83.
I accept both propositions. RCT says that the first proposition is
contrary to Prof Merges’ opinion. Accord’s submission is that it is not
because the first proposition from Fujifilm is concerned
with formalities, or rather the absence of them whereas, as Accord puts it in
its closing:
“262. […] The issue is not whether there are special
requirements for a person to be nominated, but the standard of proof necessary
to show that a person has been nominated (regardless of the means by
which that nomination occurs). This is particularly important where the method
by which the nomination is said to have occurred is by an implied contract.
There is no extra “requirement” upon them to nominate in a particular way (for
example in writing, etc.), merely to prove to the Court’s satisfaction that
there was in fact a nomination (by any means) which demonstrated the necessary
clear intent to part with the patent rights.
84.
This is an accurate reflection of the way Accord puts its case. Neither
side addressed the question whether the standard of proof is a matter for the lex
fori and I will not do so either.
85.
My findings on this point are the following. An implied in fact
contract, inferred from matters such as the conduct of the parties is capable
under US law of transferring an equitable interest. There are no special
requirements, in the sense of formalities, under US law for a person to be
nominated as the beneficiary of rights. What must be proved is a clear
intention by the parties that a person was in fact nominated. In that sense it
must be clear and unmistakable.
The evidence
86.
RCT’s case is based on witness statements from Professor Kohn, Mr
Reckart and Dr Krishnamoorti. Accord did not seek to cross-examine the latter
two witnesses at all. At the pre-trial review Accord raised the question of
cross-examination of Professor Kohn. I refused to permit Accord to
cross-examine the witness on case management grounds. At that stage Accord’s
challenge to priority was much broader than the single point now taken.
Importantly Accord only advanced one reason why they wanted to cross-examine
the professor; it was about his awareness in 1997 of a policy document from the
university. That did not justify cross-examination and so it was refused. The
point on Professor Kohn’s awareness of a policy document has nothing to do with
the issue I now have to decide.
87.
Professor Kohn’s general evidence comes down to the following:
i)
He does not recall having, or signing, any formal employment contract on
joining Houston or subsequently.
ii)
Throughout the FAA Project, Professor Kohn understood that he would not
own the rights in his inventions, nor any patents in those inventions.
Moreover, he understood he would assign any rights to RCT if required by Houston.
iii)
In return, upon commercialisation of any of his work by RCT, Professor
Kohn and Houston (together with members of his laboratory group) would receive payment
from RCT.
iv)
The work described was all part of a single project, the FAA Project. The
various strands of work, including the work on lacosamide, all fell within the
FAA Project.
v)
Professor Kohn understood that the arrangement between Houston, RCT and
himself was the same for lacosamide as it had been agreed to be for all the
other work within the FAA Project. He started the work which led to the FAA
project in 1973. Six patent applications were assigned to RCT under the FAA
project before lacosamide.
88.
Pausing there, two matters can be mentioned. One relates to paragraph
(ii). Professor Kohn’s witness statement is clear in this respect that he
understood he would assign to RCT if required by Houston. A question is
whether he ever was required by Houston to assign lacosamide.
89.
The other matter is that RCT submitted that these points and point (iv)
in particular meant that it was therefore a reasonable assumption of any party that
the same arrangements that had applied to the previous work within the FAA
Project would apply equally to the lacosamide work. It depends what you mean.
I accept that lacosamide was governed by the terms which governed the FAA
project, in effect the 1966 agreement. However what is clear, as Accord
submitted, is that the proper process required a decision by Houston to offer
to RCT (and so on) as was followed for other inventions in the FAA project.
There is no basis for assuming that a previous decision by Houston to offer a
particular invention made in the FAA project (i.e. a particular set of
compounds) applied to another different invention made later in the same
project (i.e. lacosamide). Moreover there is no evidence that Professor Kohn
made such an assumption on that basis. He does not say that is what he did.
90.
Turning to lacosamide specifically, Professor Kohn’s evidence is that in
1993 he identified twelve FAA compounds (including lacosamide) as worth
testing. He does not mention the work reported in the earlier Le Gall thesis
but nothing turns on that.
91.
No invention disclosure document is now in the possession of RCT.
Professor Kohn explains he told RCT about lacosamide (and the corresponding
racemate) in correspondence. He used the same FAA project number as before.
There were pharmacological tests run on the compounds at an organisation called
NINDS which is in effect the NIH. There is evidence of discussions between the
professor and RCT in 1993/94 about RCT being authorised by the professor to
discuss those results with NINDS. Professor Kohn’s evidence about this is
clear that he was working on the basis that RCT were responsible for patenting
these compounds. What is not said is why. There is no reference to Houston
having made a decision to offer the compounds to RCT. A fair reading of this
evidence is that Professor Kohn simply assumed that RCT were responsible for
patenting all FAA compounds.
92.
By October 1994 Professor Kohn had obtained “neat test results” from the
NIH for lacosamide. That expression comes from a letter from him to RCT which
discussed RCT taking action on licensing. Neither this letter nor the previous
letters show on their face that they were copied to Houston. That of course
does not mean they were not.
93.
An issue arose in early 1996 to delay publication of Daeock Choi’s PhD
thesis pending filing of the first priority application in March. Professor
Kohn wrote to Dr Bear, the Dean of the College of Natural Sciences and
requested the delay, as he put it to Dr Bear because “it is very important to
protect the university’s interests”. Publication was delayed and the priority
application was filed by Professor Kohn in March 1996 with the assistance of
RCT.
94.
A week later Professor Kohn wrote to Dr Robbins, the Director of
Technology at Houston because RCT had made a payment under the FAA project.
The letter was concerned with identifying which members of the Professor’s
group were entitled to a share. It is not clear what exactly this payment was
for. I do not accept the suggestion (if made) that this was a stage payment
arising from a decision by RCT to accept an offer of lacosamide by Houston.
95.
On 4th February 1997 Professor Kohn completed the assignment in favour
of RCT which has been referred to already. In his statement Professor Kohn states
that he felt that assigning his rights in the lacosamide invention made sense
to him in light of the relationship between Houston and RCT, and the manner in
which previous FAA inventions had been dealt with. This makes sense but note
that here the Professor is referring to assigning his own rights to RCT and not
to assigning Houston’s rights.
96.
Professor Kohn refers to the FAA project as a whole, the fact that
payments were made by RCT, and that there was regular communication and annual
reports from RCT. He says he kept in close contact with Houston about the
inventions and that he took care to keep Houston informed of his dealings with RCT.
The contacts included telephone calls as well as carbon copies of letters.
Professor Kohn says that all three parties (Professor Kohn, Houston and RCT) were
in contact throughout and Houston never once suggested that they were anything
other than fully content with Professor Kohn’s co-operation with RCT pursuant
to arrangements described. He said that Houston never once suggested to him that
it did not fully endorse his passing of information about the FAA inventions to
RCT or assigning his rights in these inventions to them.
97.
Professor Kohn states that one of his relevant contacts at Houston’s Office
of Sponsored Research and Technology Transfer Department was Mrs Judy Johncox.
That matters because (and I find) that department would be the correct part of
the university to make a decision to offer an invention to RCT and because (and
I find) Mrs Johncox would be one of the relevant individuals to participate in
such decision making. I will return to Mrs Johncox below.
98.
The professor ends his witness statement by summarising that his
understanding was that he would not own the rights in the inventions but that
he would assign them to RCT in accordance with Houston’s wishes.
99.
Turning to Dr Krishnamoorti’s evidence, this can be addressed much more
briefly. He was not at Houston until well after the relevant events. He
explains that lacosamide is the most successful technology transfer undertaken
by Houston and has earned the university over $50 million to date with more to
come. He is not aware of any disputes about ownership arising out of the FAA
project or any suggestion that Houston does not or did not consider that RCT
was entitled to the rights it has. He says that based on current practice,
which as far as he is aware is the same now as it was then, either Houston
would have assigned the intellectual property to RCT or to the extent that
Professor Kohn had not executed an assignment in favour of Houston, Houston
would have required Professor Kohn to assign their interests directly to RCT.
100.
Mr Reckart’s evidence explains the origins of RCT. He was RCT’s general
counsel from 1986 to 2001 and therefore can speak from first hand experience.
He explains how the FAA project ran and explains the usual procedure of
notification to RCT of the inventions and acceptance. In this respect
(paragraphs 23-35) he is talking about the time before lacosamide.
101.
In relation to lacosamide Mr Reckart’s evidence deals with the same
correspondence between Professor Kohn and RCT in 1993/94 discussed by Professor
Kohn. In relation to the filing of the priority document by Professor Kohn in
March 1996 with RCT’s assistance, Mr Reckart points out that as part of that
filing a declaration was filed which he signed on 8th March 1996.
The declaration is about the “small business” status of RCT. It states that
the rights in the invention have been conveyed to RCT. The form includes a
statement that the signing person is aware that willful false statements are
punishable by a fine or imprisonment. The timing of this declaration implies
that if there had been a decision by Houston to offer the invention to RCT and
an acceptance of that offer by RCT, it most probably happened before 8th
March 1996.
102.
Mr Reckart refers to an agreement for a joint collaboration signed in
September 1996 by Houston, RCT and Queens University of Kingston, Ontario,
Canada. He says that this arose following Professor Kohn’s invention of
lacosamide and was to look into new CNS active agents. On behalf of Houston
the agreement is signed by Dr Arthur C. Vailas, the Vice Provost of Research.
The agreement records that RCT has patents arising from inventions made at
Houston and gives a project number which is the FAA project number. There is
no express reference to lacosamide.
103.
On 10th January 1997 Mr Reckart wrote to Ms Johncox at
Houston (copied to Dr Vailas). The letter refers to the priority application
from a year before, that is the one which includes lacosamide, and informs her
that a PCT application was going to be filed. I am not aware of any reply to
that letter. RCT emphasises that Ms Johncox was in the department of the
university which would make or would have made a decision to offer inventions
to RCT. That would also explain why Ms Johncox is the person to whom Mr
Reckart was writing about patent applications.
104.
In relation to the February 1997 assignment, Mr Reckart states that in
his view it is consistent with the 1966 agreement whereby the university may
recommend to faculty members to assign their inventions to RCT. I accept that
the assignment is consistent with the agreement (see clause 3 which is
addressed below).
105.
Mr Reckart explains that RCT kept Houston up to date on patent
prosecution after this.
106.
Finally, in closing RCT produced a disclosure list from which it
submitted that one can infer many documents from the relevant period have been
lost.
Inferences
107.
That concludes my summary of the evidence. RCT submitted I should infer
from this evidence that the procedure under the 1966 agreement had been
complied with and therefore Houston had decided to offer lacosamide to RCT and
the Professor’s assignment to RCT had been done because Houston had required
him to do it. Alternatively I should infer the existence of an implied in fact
contract effective to assign the equitable interest. Finally and in any case I
should find that RCT were in the position of a bona fide purchaser for value
without notice and so took good title under the 1997 assignment even if Houston
still had equitable title at the time it was executed.
108.
Accord submitted that the principles identified by the Court of Appeal
in Wisniewski v Central Manchester Health Authority
(unreported 1st April 1998 Roch, Aldous and Brooke LJ) and by the
Supreme Court in Prest v Petrodel [2013] UKSC 34 meant
that the court was precluded from drawing the relevant inferences.
109.
The leading judgment in Wisniewski was given by
Brooke LJ, which whom Roch and Aldous LJJ agreed. After citing a number of
authorities including Herrington v BRB [1972] AC 877 the
judge summarised the principles as follows:
"(1) In certain circumstances a court may be entitled to
draw adverse inferences from the absence or silence of a witness who might be
expected to have material evidence to give on an issue in an action.
(2) If a court is willing to draw such inferences they may go
to strengthen the evidence adduced on that issue by the other party or to
weaken the evidence, if any, adduced by the party who might reasonably have
been expected to call the witness.
(3) There must, however, have been some evidence, however
weak, adduced by the former on the matter in question before the court is
entitled to draw the desired inference: in other words, there must be a case to
answer on that issue.
(4) If the reason for the witness's absence or silence
satisfies the court then no such adverse inference may be drawn. If on the
other hand there is some credible explanation given, even if it is not wholly
satisfactory, the potentially detrimental effect of his/her absence or silence
may be reduced or nullified.”
110.
Accord then referred to paragraphs 43-45 of the judgment of Lord
Sumption in Prest v Petrodell. In the first paragraph
Lord Sumption explains that what is in issue depends on what presumptions may
properly be made against a husband given that the defective nature of the
material is down to his own persistent obstruction and mendacity. There are
then references to Herrington, to R v IRC ex
parte Coombs [1991] 2 AC 283 and to Wisniewski.
Subject to an irrelevant caveat about matrimonial cases, Lord Sumption states
the principle in paragraph 44 as being that:
“There must be a reasonable basis for some hypothesis in the
evidence or the inherent probabilities, before a court can draw useful
inferences from a party’s failure to rebut it.”
111.
The reliance by Accord on Prest v Petrodell and on
some of the statements in Wisniewski seeks to portray
Professor Kohn as an absent witness or to portray RCT as a litigant who elected
to call no relevant witnesses. That would be unfair. I have addressed the
pre-trial review already.
112.
Nor is it really fair to say that any of RCT’s witnesses are “silent”
given their extensive evidence directed to the issues. However the real point
Accord is making is that Professor Kohn does not state in terms that he
disclosed lacosamide to Houston first before disclosing it to RCT, nor that
(regardless of when his disclosure to Houston took place) in any event the
university actually did make a specific decision to offer lacosamide to RCT and
then directed Professor Kohn to execute an assignment in RCT’s favour. None of
the other witnesses say that either. Therefore, the argument goes, given this
absence of evidence, the court should not infer that something happened when
the witnesses do not say that it did.
113.
I have no doubt that from his own point of view, Professor Kohn at all
times did what he did because he thought he was obliged to the university to do
so and because he thought the university thought he should. However reading
Professor Kohn’s evidence as a whole, it seems to me that by the time
lacosamide was identified he also thought or assumed that the university was
obliged to assign any FAA invention to RCT. The heart of the problem is that
this has not been established (and I find would not be correct). However this
assumption on the professor’s part may explain where the problem arises.
114.
I am not satisfied I can draw the inferences of fact necessary to
support RCT’s primary case that the procedure under the 1966 agreement had been
complied with before 4th February 1997. This depends on what did or
did not happen as between Professor Kohn and the university. For example one
approach which RCT’s case invites me to take is to infer that Professor Kohn
must have completed an invention disclosure document for lacosamide for
delivery to the department in Houston in order for them to make the decision
whether to offer that invention to RCT and for the document to then be passed to
RCT. The fact such a document is not available in disclosure today from either
RCT or Houston must therefore be simply because it has been lost. But
Professor Kohn does not say that is what did happen nor does he even say that
it is what must have happened albeit he now has no recollection of it. The
most he says is that RCT does not today have any formal invention disclosure
document but he does not even state in terms that such a document ever
existed. He is simply silent on that point. In my judgment it would be wrong
to infer that such a document must have existed but has been lost, without at
least some statement to that effect by a witness or in a disclosure list. RCT
referred to a list of what has been disclosed in closing but that list does not
help. It does not, for example, include an invention disclosure document as a
document which did exist but has now been lost.
115.
The same problem arises with the other inferences necessary to support
this part of the case. I cannot infer that the university made a decision to
offer lacosamide to RCT. Neither Professor Kohn or Dr Krishnamoorti say that
is what happened nor, again, does Professor Kohn say that it must have happened
albeit he has forgotten. All Professor Kohn does is give generalised evidence,
which I accept, that the university was aware of what he was doing. That does
not mean that they had made a specific decision about lacosamide. The same
problem also arises for an inference that the university instructed Professor
Kohn to execute the assignment on 4th February 1997. He does not
say that that is what happened.
116.
Similarly I cannot make the findings necessary to infer the existence of
an implied in fact contract to assign the equitable interest. That fails for
the same reasons. There is nothing from which to infer that the university
actually did intend that the lacosamide invention should be patented by RCT.
There is no evidence the university thought about it at all.
117.
I find that on the date Professor Kohn executed the assignment to RCT,
he held the bare legal title to the invention (including the priority right)
but Houston held the beneficial or equitable title. The Professor thought he
was doing what he was supposed to do in executing that assignment but the
assignment was not an assignment of Houston’s equitable interest.
118.
RCT made a submission based on agency but I do not see how that would
work on the facts in this case.
119.
I turn to the bona fide purchaser argument. Professor Chisum explained
that in the USA the common law concept of a bona fide purchaser for value
without notice had been codified in statute at 35 USC §261. This was not in
dispute. The only issue is notice.
120.
Here attention switches from Houston to RCT. RCT clearly knew what the
terms of the 1966 agreement were. Mr Reckart does not say that RCT received a
formal offer of lacosamide from Houston directly. Accordingly for the same
reasons as before I decline to infer that that must have happened. However the
invention was clearly disclosed to RCT and by the time Professor Kohn filed the
priority document in March 1996, RCT believed it was entitled to the invention
(see Mr Reckart’s declaration to that effect), albeit a formal assignment would
be needed to perfect its title. That assignment came in February 1997 and Mr
Reckart explains that he considers that the terms of the assignment are
consistent with the 1966 agreement under which Houston may recommend to faculty
members that they assign their inventions to RCT.
121.
Mr Reckart’s evidence here is supported by the terms of the February
assignment itself. Clause 1 notes that the inventor is obliged to assign his
rights in the application to Houston or its designee. Clause 2 describes what
is the 1966 agreement and states that RCT has evaluated the invention, is now
attempting to commercialise it and is obliged to pay Houston a share of the
revenues. Clause 3 states in terms that the inventor wishes to assign his
interests to RCT in furtherance of his obligations to Houston and Houston’s
obligations to RCT. Thus although the document states in terms it is an
assignment of Professor Kohn’s interest (rather than, say, any interest of
Houston’s) the document also makes clear that this is the inventor assigning to
RCT as Houston’s designee and pursuant to Houston’s obligations to RCT. That
must be on the footing that the invention has already been offered by Houston
to RCT and has been accepted by RCT.
122.
From RCT’s point of view the February assignment was the assignment RCT
would be expecting to perfect its title. Its context was that shortly
afterwards RCT would be filing an international application under the PCT.
Also relevant is the 10th January 1997 letter from Mr Reckart to Ms
Johncox about the forthcoming PCT application.
123.
Accord submitted that the circumstances and the recitals in the
assignment were such that RCT were on notice that they at least needed to
enquire about the university’s rights. I reject that submission. The evidence
works the other way round. All the indications available to RCT were that the
university was aware of what was going on and that Professor Kohn was executing
the assignment because he was obliged to do so pursuant to his obligations to
the university. Those indications do not only derive from Professor Kohn but
also from the university itself, by Dr Vailas’ signature on the Queen’s
University contract in mid 1996. While that document does not name lacosamide,
it is concerned with the FAA project for which the patents were being
prosecuted by RCT and, from RCT’s point of view, by then that included
lacosamide. I find that on 4th February 1997 RCT was not on notice
of any possible conflicting interest held by the university. Therefore RCT
acquired good title to the invention including any priority right. Any
equitable interest in the invention which the university did hold prior to the
assignment was destroyed or overridden by the assignment to RCT.
124.
Accordingly RCT had the substantive priority right when the PCT
application was filed.
Obviousness
125.
The only point of law arising relating to obviousness is the idea of
“supplementary information”. This term was used by Accord to refer to
information which was not common general knowledge but which would be obvious
to acquire. Recent cases on this are KCI Licensing v Smith &
Nephew [2010] EWCA Civ 1260 at paragraph 6 (endorsing Arnold J at
first instance [2010] EWHC 1487 (Pat)) and Richter v Generics
[2014] EWHC 1666 (Pat) (Sales J) upheld at [2016] EWCA Civ 410. KCI
states the principle that that sort of information can be relevant and Richter
is a case in which it led to a finding of obviousness. Mind you there is a
notable contrast on the facts between this case and Richter.
In Richter the supplementary information was a single fact
(that something reported as 1.5g was actually 1.5mg). In this case Accord rely
on the distillation of what is said to be an obvious literature search.
Le Gall
126.
Le Gall is a Master’s thesis, describing work undertaken under the
supervision of Professor Kohn in the Chemistry Department at the University of
Houston. The study describes work carried out on functionalised amino acids
(FAA), both synthesising them and evaluating them in the MES test for their
anticonvulsant drug potential.
127.
Le Gall built on work already carried out by the Kohn group. He had two
compounds around which he sought to develop a SAR, both of which were
previously discovered by the Kohn group. The base structure used by Le Gall is
shown below, with the variable R group in blue:
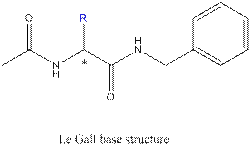
128.
Le Gall only modified the R side chain. The starred (*) carbon is a
chiral carbon atom.
129.
The two starting compounds used by Le Gall are derivatives where the R
group is a methyl or a phenyl respectively. The compounds are referred to in
Le Gall as 68a and 68b:
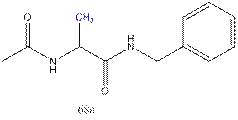
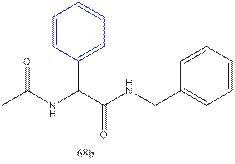
130.
In his work Le Gall produced three different sets of derivatives, each
set with related but different R groups; one set numbered 69a-h, one numbered
86a-b (which were intermediates in the synthesis of 69a-h but were still tested
for activity) and the other numbered 107a-e. Some data for other compounds, not
made by Le Gall, are also given and addressed in the thesis.
131.
Following an abstract which summarises the work, the thesis is split
into three sections: an introduction, and Chapters I and II. The introduction
summarises the state of research into anticonvulsant drugs at the relevant date
and the previous work of the Kohn group. At page 42 (using the document’s own
page numbers) the following passage appears:
“Recently, Kohn and co-workers68,102,103 described
the anticonvulsant properties of several N-benzyl amino acids. Compounds 68
contained many of the structural elements
(i.e. 46b, 46c) present in phenytoin (13a) and
the benzodiazepines (24). Recent evidence has indicated that these compounds
possess an unique mode of action, suggesting that they may be a new class of
anticonvulsant drugs.68 Interestingly, the D-enantiomer of 68a was
thirteen times more active than the L-isomer when tested orally in mice in the
MES seizure test. A comparable difference in activity was also noted for the
two stereoisomers of 68b. This
information coupled with the stringent structure-activity relationship observed
for this class of compounds68 has led to the speculation that the
anticonvulsant properties of these compounds may be related to interactions
with specific receptor sites.”
132.
The base structure set out above is then shown in the thesis with
compounds 68a and 68b shown (along with 68 c, d and e). Accord places emphasis
on the comparison to phenytoin, the reference to a possibly unique mode of
action and the statement about the relative potency of enantiomers. On the
last point one needs to take care not to mix up the difference between the
relative potency of two enantiomers as opposed to the potency of the racemate
as compared to one active enantiomer. Even if one enantiomer is entirely
inactive and the other is active such that the ratio of their activities is not
thirteen times but effectively infinite, the difference in potency between the
racemate and the active enantiomer in this idealised example is only half.
That would be common general knowledge.
133.
The compounds synthesised in chapter I are analogues where the R group
is replaced by various different aromatic side chains. The rationale given by
Le Gall is that these are analogues of the phenyl compound 68b. In particular
Le Gall makes and tests the following derivatives (showing the R groups):
name structure
name structure
2-furan (69a):
 2-pyrrole
(69b):
2-pyrrole
(69b):
2-thienyl
(69c):  3-
thienyl (69d):
3-
thienyl (69d): 
β-napthyl
(69e): 2-benzofuryl
(69f):
2-benzofuryl
(69f): 
3-indoyl (69g):
2-benzo-[b]thienyl
(69h):
134.
These eight compounds can be considered in two groups of compounds – the
one group of the smaller single ring structures (the first four), and the
second with larger benzo-fused ring structures (the second four). Strictly
compounds 69a and 69b should be called furyl and pyrryl above but I have called
then furan and pyrrole for consistency with the rest of this judgment and the
way in which the arguments at trial were expressed. The 2 in “2-furan” refers
to the point in the furan or pyrrole ring which is attached to the backbone of
the structure.
135.
The pharmacological evaluation of these compounds begins at page 102,
and the data are shown on pages 104-105. The values obtained are described as
having been generated using the MES test to create the ED50’s, with the
compounds first being administered to groups of four mice at 300, 100 and 30
mg/kg, and then effective compounds being further tested in groups of 12 mice to
determine an ED50. The TD50’s were generated using the horizontal screen test.
136.
Compounds 69a and 69b have ED50’s of 10.3 mg/kg (with 95% confidence
limits of 9.1-11.6 mg/kg) and 16.1 (with 95% confidence limits of 13.2-19.9
mg/kg) respectively. The TD50 values are also given for these compounds with
69a having a TD50 of about 40, and 69b having a TD50 of 30-100.
137.
In Chapter I Le Gall also shows data for a series of other compounds,
some synthesised by Le Gall as intermediates, and others whose data are gathered
from other members of the Kohn team. These are numbered 68c, 68d, 86a and 86b.
The compounds are the following derivatives (showing just the R groups):
name structure name structure
Methoxy (86a)  Ethoxy
(86b)
Ethoxy
(86b) 
2-methylthio
ethyl (68c) Iso-propyl
(68d)
Iso-propyl
(68d) 
138.
These compounds were not as highly active, with the ethoxy (86b) showing
the highest activity of 62.0 mg/kg. The activity of the methoxy derivative
(86a) was 98.3 mg/kg.
139.
Chapter II is concerned with a series of analogues of the methyl
compound 68a. Several of the compounds in this group were unsaturated and had
a polar substituent at the α-position (page 132). The five compounds are
107a-e. The first four were made and tested. Showing the R group only they
are:
Cyano (107a) 
Amido (107b) 
Ethylcarboxylate
(107c) 
Hydroxymethyl
(107d) 
140.
Compounds 107a, 107b and 107C have ED50s in the MES test of >300
mg/kg while 107 has an ED50 of “>100, <300” mg/kg. The TD50s for 107a
and 107b are >300 mg/kg while the TD50 for compounds 107c and 107d are
<300 mg/kg.
141.
The fifth compound 107e was made but never tested. This has the
following structure:
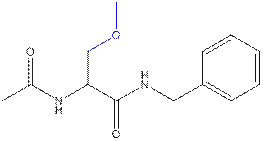
142.
This is the structure for which lacosamide is the R enantiomer.
Compound 107e is the racemate.
143.
Le Gall discusses these compounds at p153-155 as follows:
“Compounds 107a-c did not exhibit significant activity in the
MES seizure test. The lack of anticonvulsant properties of these adducts was
interesting in light of the pronounced activity of the methyl analogue 68a. A
tentative explanation for this dichotomy of results can be offered. In a first
approximation compounds 68a and 107a-c all contain relatively small
substituents. The primary difference between the two sets of compounds is the
presence of an electron-donating (68a) or an electron-withdrawing (107a-c) moiety
at the α-carbon. Our previous studies have indicated that the activity of
the drug candidate in the MES seizure test is enhanced by the presence of
electron-donating groups at the a-carbon. The negligible activity of 107a, b
and c is in agreement with this trend.
The serine derivative 107d exhibited only slight
anticonvulsant activity in the MES seizure test. The activity of this compound
was considerably diminished from the corresponding isomeric methoxy ether 86a (Table
33). This result may reflect the inability of 107d to readily pass through the
blood-brain barrier. The more lipophilic methoxy ether 107e has not been
evaluated yet. The close structural analogy of this compound with 86b suggest
that this adduct may have good anticonvulsant activity.”
144.
In this passage Le Gall seeks to explain the weak activity of the
compounds tested and draws an analogy between 107e and the compound 86b which
had been tested. Note that the activity of the ethoxy compound 86b is 62.0
mg/kg and that provides an indication of what the author regards as good
activity (in 1987).
145.
The thesis ends with general conclusions at p164-165. Amongst other
things the conclusions draw attention to the activity of the five membered ring
heteroaromatic analogues of 68b (such as the furan 69a and pyrrole 69b) and
describe their activities as being similar to phenytoin and diazepam. The idea
that the enantiomers may be more active than the racemates is mentioned. There
is then a reference to stringent electronic and steric requirements and the
statement that the study supports the notion that the α-carbon substituent
interacts with an electrophilic site on the receptor. Various suggestions for
future work are made. None of the suggestions are about doing anything with
compound 107e.
What to do with
Le Gall
146.
Accord no longer rely on a case that the claimed invention is obvious
over Le Gall alone. I am not surprised. Assume the skilled team thought it
was worth considering taking compound 107e forward at all, despite the quality
of Le Gall’s reasoning and despite the poor performance of 107 a-d; that would
be based on the last two sentences of the passage from p155 quoted above which
links the possible activity of compound 107e to the activity of compound 86b.
Given Le Gall alone and the common general knowledge the best that could be
said in terms of expectation of success based on this, is that the activity of
the 107e racemate might be comparable to compound 86b, which was 62.0 mg/kg.
The thesis was written in 1987 but by 1996 that level of activity would not be
good enough to make it obvious to try with a fair prospect that a useful
anticonvulsant drug might be the result. For the argument to have any traction
considering a skilled team in 1996, the expectation would have to be of a much
lower activity than that. Even halving it, based on the best you can get from
Accord’s racemate/ enantiomer argument, it is still not worthwhile.
147.
Accord’s case is that given the Le Gall thesis in 1996, the first thing
the skilled team would do is a literature search into the work of the Kohn
group. I agree that would be the conventional thing for a skilled team to do
given Le Gall in 1996 if they wanted to do anything at all. Although RCT
contended that the skilled team having read Le Gall in 1996 with interest would
then decide they were not interested, I do not accept that. I find that the
skilled team in 1996 would find the chemical reasoning in Le Gall
over-simplified and unconvincing but the pharmacological data on the compounds
tested is sufficiently interesting for a skilled team to do the next
conventional thing, which would be a literature search. The search would
include searching using the named authors. The thesis is clearly the product
of work in Prof Kohn’s group at the University of Houston and the names Kohn
and Le Gall would be searched as well as other names appearing such as Conley
(Conley papers are cited by Le Gall). The thesis also shows that the major
pharmaceutical company Eli Lilly have been involved and the names Leander and
Robertson would be seen in the thesis as linked to Lilly (see the
acknowledgement). The conventional search would also involve not only
searching by author name but also searching by reference to some of the
compounds such as the furan.
148.
The team would find many papers. I have not had my attention drawn to a
single comprehensive list of the material which would emerge from this
exercise. It would include the following:
i)
Functionalized DL-Amino
Acid Derivatives. Potent New Agents for the Treatment of Epilepsy by Judith
Conley and Harold Kohn, published in J Med Chem 1987 Vol 30 567-574 (“Conley
1987”)
ii)
LY274959 - A Potent, Stereoselective Anticonvulsant against Maximal
Electric Shock (MES) Seizures by Leander et al published as an abstract
submitted to the Princeton Drug Research Symposium on Current and future trends
in anticonvulsant, anxiety and stroke therapy May 21-23, 1989 – Princeton NJ
(“Leander”)
iii)
Preparation and Anticonvulsant Activity of a Series of Functionalised
α-Aromatic and α-Heteraromatic Amino Acids by Kohn et al
published in J Med Chem 1990 Vol 33 pp919-926 (“Kohn 1990”)
iv)
Preparation and Anticonvulsant Activity of a Series of Functionalised
α -Heteroatom-Substituted Amino Acids by Kohn et al published in J Med
Chem 1991 Vol 34 pp2444-52 (“Kohn 1991”)
v)
Synthesis and anticonvulsant activities of α -heterocyclic
α -acetamido-N-benzylacetamide derivatives by Kohn et al published in
J Med Chem 1993 Vol 36 pp3350-60 (“Kohn 1993”)
vi)
Anticonvulsant Properties of N-Substituted α,α-Diamino Acid
Derivatives by Kohn et al published in J Pharmaceutical Sciences 1994 Vol
83 pp689-91 (“Kohn 1994”)
vii)
Synthesis and Anticonvulsant Activities of
α-Acetamido-N-benzylacetamide Derivatives Containing an Electron-Deficient
α-Heteroaromatic Substituent by Patrick Bardel et al published in J
Med Chem 1994 Vol 37 pp4567-71 (“Bardel).
149.
There are more papers from the Kohn group than this and the conventional
literature search by the skilled team would turn up more papers than the seven
listed above but I find it would include those seven. Conley 1987 is referred
to in the Le Gall thesis and would be obtained even though it predates Le
Gall. Prof Cox’s own searches conducted for the purpose of this case did not
turn up the Leander abstract but it was published and available and I find that
in 1996 it would be identified.
150.
Conley 1987 is reference 68 in Le Gall. It is the reference for the
statement that the new class of compounds have a unique mode of action in the
passage quoted above from p42 of the thesis. It was put to Professor Löscher
that if the skilled team followed this up they would read Conley 1987 and see
the profile of activities in the various pharmacological assays in that paper
of two compounds mentioned in Le Gall. The compounds are 68a (methyl) and 68b
(phenyl) in Le Gall while in Conley 1987 they are 1a and 1d. In
cross-examination a table was produced of those results to show the activities
of these two. The tests are all in mice. They are the MES test, four tests
using chemical convulsants (sc Met, sc Bic, sc Pic and sc Strych) and the
neurotoxicity rotarod test. Professor Löscher did not agree with the way Le
Gall had put it based on these data. Professor Löscher’s way of expressing
what would emerge from this was that the compounds had a different
pharmacological profile in seizure models from known antiepileptic drugs,
although he thought the phenyl compound (68b) was quite similar to phenytoin
and would be more confident that the methyl compound (68a) was potentially
interesting.
151.
The pharmacologist and the medicinal chemist would review the results of
the literature search as a whole. The team would see the work that the Kohn
group had done over an extended period of about ten years in this class of
compounds. A large number of compounds with the same basic structure have been
synthesised and tested. The vast majority are different substitutions at the R
position described above in what I have called (but they did not) the Le Gall
base structure. There are some substitutions elsewhere too. I am not aware
that they have been counted but there must be well over fifty different
compounds spread over about ten years, all of which were new chemical entities
and were synthesised and tested at least in the MES and neurotoxicity tests.
That is a huge body of pharmacology and medicinal chemistry work.
152.
If the team was going to take things forward they would look for the
best compounds arising from this work. There are two ways of looking at this.
One is the Leander Abstract. The authors are from Lilly and the Kohn group.
As an abstract it is very brief. Its compound is called LY274959 (LY obviously
refers to Lilly). It is the R enantiomer of the furan compound (Le Gall’s
69a). The abstract shows that this compound has been taken further down the
development process than many of the others and is showing promising results.
Leander reports MES and neurotoxicity (horizontal screen) test results both ip
(intraperitoneal) and po (oral) in mice and po in rats as well as some chemical
convulsant results in mice (sc Met, sc Bic and sc Pic). The conclusion is that
“LY274959 has a preclinical profile that suggests it would be a
therapeutically useful anticonvulsant in humans.” The skilled team would
be very interested in that despite the limitations in the detail of the
information available.
153.
So one obvious thing for the team to do could be to work on LY274959 and
take it forward. But that is not the only thing a skilled team might do.
154.
In his evidence Professor Löscher had pulled together a table of the
compounds from this material which had ED50s in the MES test of about 10 mg/kg
or lower. I find that the skilled team (really the pharmacologist) would pull
together a list of what they thought were the best compounds. Of course that
is not all that would come out of the significant work of reviewing all these
papers but producing such a list would be conventional.
155.
An expanded version of Professor Löscher’s table (expanded not to add
compounds but to add more data) is the following:
Table 1: Kohn group compounds
with potential MES ED50 values < 10 mg/kg
(I have edited
Professor Löscher’s table in two further ways: the reference to LY274959 is
added to (R) -2g and the entry for (R,S)-11 is edited to reflect the range of
TD50 values rather than expressing it as a footnote as in the original table.)
156.
Note that Professor Löscher’s table includes two entries for phenytoin.
They show slightly different MES ED50s. In reality the team would probably
have more comparator compounds, not just phenytoin, and put them elsewhere but
nothing turns on that. In this table compound (R)-2g is the R enantiomer of Le
Gall’s furan compound 69a. In other words it is LY274959.
157.
Now before going any further a point arises on the quality of the data.
A conventional skilled team would not make major decisions based simply on reported
data. It would not be practical to rerun the whole of the Kohn group’s work
but they would seek to reproduce any results on which they were going to place
any significant weight. Repeating all the results just in this table would be
very substantial albeit routine work. It would not just involve synthesising
and testing the compounds but running various internal standards and controls.
For the purpose of the analysis I will assume the repetition of results would
be identical, although that is not really a credible assumption given these are
tests in a biological system and using a small number of subjects.
158.
For the team to move forward from this sort of information would involve
the team taking a phenotypic screening approach to drug development rather than
a target based approach or one based on modifying an existing clinically
approved compound (such as Professor Cox’s work on lamotrigine). If Professor
Löscher was involved personally I doubt that is the approach which would have
been taken but in 1996 phenotypic screening was one of the conventional
approaches taken by skilled teams.
159.
Professor Cox’s evidence was that 3n and 3l would stand out as the most
active. I do not accept that the skilled team’s approach would be quite as
simple as that. Professor Cox is a medicinal chemist albeit with a wealth of
experience in liaising with pharmacologists. The pharmacologist member of the
skilled team would be familiar with the various tests such as the MES test.
They would not attribute much significance to small differences in the test
results reported in this way. Professor Löscher’s evidence was that this sort
of early phase phenotypic screening is relatively crude and uses small cohorts
of animals. He said the pharmacologist would not make fine distinctions
between ED50 values in the MES test and gave an example that no distinction
would be drawn between values of approximately 10 mg/kg and 25 mg/kg. The
point of the example was a relative one.
160.
Nevertheless the skilled team taking the approach of moving forwards
from a table of best compounds would indeed be interested in 3n and 3l from the
above table but also in other compounds. I doubt they would dismiss any of the
nine compounds in Professor Löscher’s table. To a pharmacologist an ED50 of 6.2
in the MES test conducted this way is not materially different from an ED50 of
10.4 (and that is irrespective of 95% confidence intervals).
161.
Compounds 3l and 3n are racemates. Accord contends that this means the
team would expect the activity to be in a single enantiomer and that enantiomer
(most likely R rather than S given the other data) would have an ED50 value
twice as good as the value for the racemate – so 6.3 mg/kg for compound 3l
would be expected to be just over 3 mg/kg. That is too simplistic. The
skilled team would expect the activities of the two enantiomers to differ and
expect that the useful activity would probably reside in one of the
enantiomers, although it may not and both enantiomers may have relevant effects
(one may even have a negative effect). They would think it probable that the
more active enantiomer had a better ED50 than the racemate to some degree (but
it might not) but they would not think it was reasonable to expect the active
enantiomer to have twice as good a value as the racemate. The same racemate
point also applies to compounds 13, 18, 11 and 19. Note that compound (R)-30
is a single enantiomer. It differs from LY274959 in that it has a fluorine on
the benzene ring (shown in the right of the Le Gall base structure above).
162.
So the racemate point is relevant but it does not go as far as Accord
contends.
163.
Now the next step involves analogues. It was common ground that the
development program is a kind of funnel, starting with many compounds, coming
up with a lead and some back up compounds and gradually coming down to fewer
and fewer as the project advances. The team would be interested in thinking
about analogues of whatever lead compounds it chose. The team would also know
from the Kohn work that a wide variety of compounds which can be regarded as
analogues of whatever compounds the team selected had already been tested.
164.
Thinking about analogues the focus turns away from the pharmacologist
and towards the medicinal chemist. Accord’s case is that at this stage the
skilled team would identify a few lead compounds from this work. They would be
or include the following three: compound LY274959 as well as compounds 3n and
compound 3l. Their structures are:
3l 3n
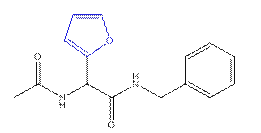
LY274959 (strictly what is
drawn is the racemate)
165.
The key question is the following. Accord contends that thinking about
analogues would take the skilled team, now led by the medicinal chemist, to
consider a methoxymethyl derivate of the base structure compounds as an
analogue worth making and testing. The compound is:

166.
This is Le Gall’s 107e. Its R enantiomer is lacosamide. Accord says
the team would test both the racemate and the two enantiomers (expecting the R
enantiomer to be the good one). I will refer to it as lacosamide although of
course that name was not used at the time.
167.
Accord’s case is that once the team had conceived of what is compound
107e the team’s expectations about its activity in tests would be a sufficient
expectation of a good result, in the standard screening tests (MES and
neurotoxicity), that claimed invention is obvious. There is an intermediate
step in that logic in that what really has to happen is the team tests the
racemate, likes it, and then tests the enantiomers and finds lacosamide.
However for the purposes of analysis this intermediate step is not a major
issue. I find that if the team did test the racemic compound 107e in the standard
screening tests (MES and neurotoxicity) they would find a good enough ED50/TD50
and PI that it would then be obvious to test both enantiomers, one of which is
lacosamide, in the same sort of tests. That would be obvious to do with a fair
expectation that the useful activity which had already been identified resides
in one of the two enantiomers. Those results would in turn be good enough for
the skilled team to then make a reasonable prediction that lacosamide would be
a useful anticonvulsant medicine for the treatment of epilepsy – at least for
tonic-clonic seizures – and that is enough to invalidate the relevant claims.
This analysis of the intermediate step is fair whether all the tests are run in
mice or in rats.
168.
The issues in this case about thinking of testing compound 107e and the
relevant expectation are primarily matters of medicinal chemistry. Professor
Cox’s opinion, maintained in cross-examination, was that there were solid
grounds for expecting good activity for lacosamide based on the information
available to the skilled team and their common general knowledge. Prof Ward’s
opinion, also maintained in cross-examination, was to the contrary. Save for
one point, this is not a topic on which Professor Cox’s longer experience in
the field as at 1996 is of significance because the issue is about chemical
reasoning, not drug development programme approaches.
169.
The exception is about what was called a “reductionist” approach to
analogue design. The point is that if you look at the furan and the
methoxymethyl parts of lacosamide respectively, you can notionally cut away the
two carbons on the left of the furan ring as it is drawn above and if you do
you get the methoxymethyl compound. In fact some hydrogens need to be put on
the terminal carbon to make it methyl. One needs to be a bit careful about
this for reasons addressed below, but this step is what Accord call the result
of taking a “reductionist” approach.
170.
Professor Cox explained that this approach of cutting away and reducing
a starting compound down to the key parts was a method medicinal chemists who
trained before the 1990s had been trained in. He referred to the training
whereby very often the approach would be applied to a complex natural product
in order to identify its pharmacophore. The term pharmacophore refers to
effectively the active core of the molecule responsible for the effect you are
interested in – consisting of the functional groups which are required for
activity and their relative positions in space. Professor Cox gave an example
of the analgesic morphine to which this approach was applied over many decades
to produce numerous simpler successful drugs.
171.
Professor Ward did not disagree that this approach was taken but he did
not agree that it justified, as he put it “literally with a pair of scissors
cutting out 107e from 69a”. His reference to the pair of scissors metaphor in
cross-examination was to acknowledge the existence of the similarity Accord
relies on while he then went on in his answer to explain why he did not agree
it was a step the skilled person would take.
172.
I accept Professor Cox’s evidence, which was not challenged, that one of
the things medicinal chemists were trained to do was take a “reductionist”
approach of the kind he described, such as paring down a large complex natural
product to a putative pharmacophore. But that general technique, which perhaps
older medicinal chemists were more familiar with in 1996 than younger ones, is
different from the very specific step in the context of analogue design of
going from one analogue (the furan) to another (the methoxymethyl). Neither
analogue is a large complex natural product. I was not persuaded that the
thinking about a change of that kind would be influenced by the age or training
of a medicinal chemist in 1996. Professor Ward said that there was no step
change in how medicinal chemists had been trained and that considering shape
and electronics of the molecule, reducing complexity and having simpler
pharmacophores was what every medicinal chemist deals with. I accept that.
173.
Therefore the fact Professor Cox’s training started before the 1990s is
not a reason to give his opinion greater weight than Professor Ward’s on this
issue.
174.
However despite RCT’s case that they would not, I think one of the analogues
the skilled team would at least conceive of would be the methoxymethyl as a
racemate. That would arise from their thinking about analogues of what are
clearly the best compounds revealed by the literature search. I am not
convinced it would be conceived by cutting away at the furan, since as
Professor Ward pointed out that “cutting away” involves a significant change to
the three dimensional structure and the electronics because it removes a planar
aromatic ring with π-electrons. But I think it could well be conceived
from thinking about the compound 3l in particular. The team would have drawn
up a full list of the compounds found in the literature search. The thing
about 3n and 3l is that they are not aromatic, unlike the other compounds in the
table above and unlike the large majority of compounds the Kohn group had
tested. The team would see that a number of other non-aromatic substituents
had been tested. Results for about 20 non-aromatic compounds are reported in
the Kohn 1991 paper. They include the Le Gall compound 86a (methoxy) which is
compound 3a in Kohn 1991 and include some compounds similar to 3n and 3l such as 3k (in which R is -NHOH) and 3m (in which R is
-N(CH3)OH). However an analogue of compound 3l in which the
nitrogen in the substituent group is replaced with a carbon had not been tested
in any of the results shown in the literature search. The team would probably
have noted it was in Le Gall as compound 107e and never tested but even if they
had forgotten that, I think they would conceive of the compound as one possible
analogue to consider, for which they had no data.
175.
Bioisosterism is the idea that certain atoms or groups in a molecule can
be replaced with certain others in such a way that the molecule is not altered
very much and so the two molecules may behave the same way. It was part of the
medicinal chemist’s common general knowledge as part of analogue design.
However bioisosteric reasoning cannot be taken too far. Professor Ward pointed
out that the table of substitutions which was to be found in the current
textbook and derived from a paper by Thornber in 1979, if read literally, would
suggest that a carbon could simply be replaced with an arsenic (replacing -CH=
with -As=). The manner in which Professor Ward expressed himself about that
indicated that he did not see it as a serious suggestion.
176.
On the other hand I accept that a medicinal chemist in 1996 could regard
replacing a linkage -NH- with a -CH2- as a possible change. That is
the change which would turn compound 3l into compound 107e. They would not see
it as a change with no possible consequences but then the medicinal chemist
would not think that about any change. The question is always about what the
possible consequences might be and their likelihoods. Unlike a carbon, the
nitrogen atom has an electron lone pair. That can be responsible for
differences in the behaviour of two otherwise identical molecules.
177.
A further possible difference would be in relation to interaction
between the adjacent oxygen and the nitrogen, as opposed to the oxygen and the
new carbon atom. Professor Ward referred to an effect called the alpha effect,
which can arise when there is a nitrogen-oxygen bond. I do not believe the
skilled medicinal chemist would think the alpha effect itself was relevant to
the activity of compound 3l since the alpha effect is about forming covalent
bonds. However, it is an example of the fact that interactions between
adjacent atoms can be relevant. I find that the skilled medicinal chemist
would have in mind that changing the nitrogen to a carbon could make a
difference given the adjacent oxygen. Given the strong electronegativity of
the oxygen it might be a small difference or even a positive difference but
then again it might not be. I was not satisfied the skilled team would be
confident they could make a reasonable prediction what difference the change
would make either based on bioisosteric considerations alone (i.e. just
swapping C for N) or taking account of the adjacent oxygen as well.
178.
Professor Cox said that on the basis of the bioisosteric switch from 3n
or 3l, the medicinal chemist would expect the activity of the methoxymethyl
compound to be “the same or better” and that such a switch was very highly
likely to be successful. Similarly he said that comparing to the furan, the
skilled person would also expect the methoxymethyl compound to be very similar
in activity.
179.
In order to make such a prediction the scientist needs to have a
hypothesis, a theory which makes predictions which can then be tested. This is
the basis for a structure activity relationship or SAR. The SAR allows the
chemist to predict what the effect of a change to the structure might be in
order to enhance the properties of the compound. This is core medicinal
chemistry. The literature search would show that by 1996 the Kohn group had
two related theories for the basis on which the compounds showed good activity
- aromaticity and the effect of a β-heteroatom.
180.
Professor Ward gave evidence that looking across all the compounds found
in the literature search, the results show cases in which aromatic substituents
work and some in which they do not, as well as cases in which substituents with
a β-heteroatom work and some in which they do
not. It is also clear that the Kohn group spent by far the bulk of its energy
on aromatic substituents. So for Professor Ward the skilled team would not
have a fair expectation that 107e would give a good result in the tests.
Professor Cox did not agree. In order to grapple with this I need to take a
step back and explain the hypotheses in more detail.
181.
Aromaticity refers to the aromatic systems in many of these molecules
such as phenyl rings. They have a π-electron cloud above and below the
ring. The concept of a β-heteroatom is as follows. The oxygen in the R
group of compounds 3l and 3n is at the β position. It is called a
“hetero” atom in the context of organic chemistry because it is different from
carbon. So a furan is called a heterocyclic structure because instead of being
all carbon atoms around the ring, one of them is different. Equally a pyrrole
is a heterocycle but this time the different atom is the nitrogen.
182.
So just as the oxygen in 3l is a β-heteroatom so the oxygen in the
methoxymethyl group in 107e is also a β-heteroatom.
183.
The skilled team would see from the Kohn group’s papers that two
hypotheses were being advanced to explain the activity of the compounds. One
theory was that having an aromatic system in the R group was responsible for
good activity and another was that it was the presence of a β-heteroatom
which was responsible for good activity. Since the R group in LY274959 is a
furan, that has both aromaticity and a heteroatom. Linking the furan to the
backbone at the 2 position locates the oxygen heteroatom at the β
position.
184.
The latest paper from the Kohn group turned up by the literature search
at the priority date would be Bardel. The following explanation of Bardel is
put in the language used in this judgment. The abstract of Bardel states that
the group’s work has shown that compounds using the Le Gall base structure and
with various R groups have displayed excellent anticonvulsant activities in
mice. It then states that analysis of the SAR for this series of compounds has
shown that what makes for good activity in the MES test is “the presence of
small, electron-rich aromatic and heteroaromatic groups” in R. The
examples of that used in Bardel are Le Gall’s furan compound 69a (numbered 2 in
Bardel and marked as (R,S)-2g in Professor Löscher’s table above) and Le Gall’s
pyrrole compound 69a (3 in Bardel). The abstract continues by explaining that
three new compounds have been synthesised (11, 12, and 13) and all three have
potencies comparable to or better than phenytoin. The abstract ends by
explaining that these findings mean that the group has to “modify in part” its
previous SAR hypothesis.
185.
Compound 11 is compound “(R,S)-11” in Professor Löscher’s table above.
The R groups for the three compounds 11 – 13 are:
186.
These structures have R groups which are aromatic rings (a pyridine,
pyrazine and pyrimidine ring respectively), but Bardel explains that these
three compounds are electron-deficient aromatic analogues in contrast to the
furan and pyrrole which in Bardel’s terms are electron-rich aromatic
analogues. All five compounds (the furan and pyrrole and the three new
compounds) are aromatic and they all have at least one β-heteroatom. The
β-heteroatom in the furan is an oxygen whereas in the three new compounds
and the pyrrole it is a nitrogen.
187.
The ED50s in the MES test for new compounds 11-13 are from 8.1 – 14.8
mg/kg. Bardel points out that the group’s theory had previously proposed that
using an electron rich aromatic group for R would result in improved activity.
The significance of the good activity of electron deficient aromatic rings is
that it is not consistent with the electron rich aromatic theory.
188.
Bardel also points out that the Kohn group had also presented evidence
that “placement of a substituted heteroatom two atoms removed from the
C(α) site” gave enhanced activity in the MES test, referring to
compounds 4 and 5 in Bardel with ED50s of 44.8 mg/kg and 87.8 mg/kg. This is
the β-heteroatom theory. Compounds 4 and 5 have sulphur atoms substituted
in the ring (they are Le Gall’s thienyl compounds 69c and 69d). They are:
189.
Bardel then states that in agreement with the β-heteroatom theory,
the trend in activity as between 11, 12 and 13 was that 13 was more potent than
either 11 or 12 (because it has two β-heteroatoms).
190.
Then Bardel states that the findings indicate that “of these two
structural determinants the latter was the more important factor for
anticonvulsant activity. Consistent with this theory was the notable
protection observed for the C(α)-heteroatom adducts 24 (ED50 = 6.2 mg/kg)
and 25 (ED50 = 31.4 mg/kg) in the MES test”. The two structural
determinants referred to are the aromaticity theory (or at least the electron
rich aromaticity theory) on the one hand and the β-heteroatom theory on
the other. So this is a clear statement that the authors are proposing here
that the β-heteroatom theory is the more important structural
determinant. Compounds 24 and 25 referred to in support of this proposal are
aliphatic (that is in contrast to being aromatic). Compound 24 is the compound
in Professor Löscher’s table labelled as compound 3l, while compound 25 is the
following:
(This compound was
compound 3o in Kohn 1991)
191.
This proposal in favour of the β-heteroatom theory and the evidence
leading to it are the high point of Accord’s case. The case is that armed with
that endorsement of the β-heteroatom theory in favour of aromaticity, the
team would have a fair expectation that 107e with its β-heteroatom in a
structure which is in effect a carbon analogue of Bardel’s compound 24 (i.e.
compound 3l in Professor Löscher’s table) would have good activity.
192.
However I do not believe the argument reflects the skilled team’s
thinking about the issue. Bardel does not say that any compound with a
β-heteroatom will work and the team would not understand Bardel in that
way. The Kohn group’s work shows examples of compounds with β-heteroatoms
which do not work. They include compounds in Kohn 1991 such as 3k (-NHOH) and
3m (-N(CH3)OH).) In Le Gall’s thesis compounds 107a (-CN), 107b
(-CONH2), 107c (-COOEt) and 107d (-CH2OH) all include a
β-heteroatom and have poor or no activity.
193.
Conversely there are compounds with no β-heteroatom at all which
have reasonable activity, such as the phenyl compound, Le Gall’s 68b. Moreover
the compounds with good activity reported by the Kohn group mostly have an
aromatic ring structure. That can be seen just by looking at Professor
Löscher’s table above. So the idea of going forward with a non-aromatic
compound would be to divert from the thrust of the Kohn group’s published
work.
194.
Also one needs to take care not to equate “aromatic” with “electronic
rich aromatic”. All aromatic compounds have a π cloud of electrons, it is
just that some are more electron rich than the others. It is a matter of
degree. The new evidence in Bardel was not that aliphatic compounds could be
active, since, as the reader would understand, the papers show the Kohn group
already knew that. The new evidence in Bardel was about aromatic compounds
which worked even though they were electron deficient.
195.
Possible explanations for the failures of compounds with a
β-heteroatom to work may be based on what is called ADME (Absorption,
Distribution, Metabolism, Excretion). These are pharmacokinetic effects which
relate to what happens to the drug after it is administered to a living
system. Absorption is the process of the drug entering the systemic
circulation after it has been administered. Distribution is the dispersion of
the drug through the fluids and tissue of the body. It could include the
ability to cross the blood brain barrier. Metabolism is the conversion of the
drug into other molecules by enzymes in the body (such as the enzyme family
cytochrome P-450 in the liver and in other places). Excretion is the removal of
the drug from the body. It is not difficult to see how these effects can be
very significant. If all the drug substance is metabolised to an inactive
metabolite shortly after it has been administered and before it can act, then
no activity will show up in the test irrespective of whether the molecule could
interact with the target. Equally if the target is in the brain and the drug
cannot cross the blood brain barrier, it will never interact with that target.
196.
I reject the suggestion that Professor Cox’a opinions about this topic
were of more weight than Professor Ward’s. Consideration of ADME effects is
fundamental to drug development and both experts were well qualified to discuss
it. There were no relevant changes in that field over time. A point arose about
the presence of cytochrome P-450 in the gut as well the liver. I do not accept
that was common general knowledge in 1996 but it is a minor issue. Also
Professor Ward gave credible evidence that his work on in vitro target
based assays also involved an analogous issue in that to interpret results one
needed to take into account whether the drug had actually reached the target
concerned.
197.
It was common general knowledge that ADME can cause unexpected and
unpredictable changes in activity. All the same it was common ground that the
team might think that compound 3k (-NHOH) as well as its carbon analogue 107d
(-CH2OH) were susceptible to being metabolised owing to the terminal
hydroxyl group and that might be the explanation for the poor activity of 107d
and the lower activity of 3k relative to 3n. But then again it might not. The
available information does not include any studies on metabolism which might
show that that is what has happened.
198.
Furthermore the team would also think that replacing the hydrogen of the
hydroxyl with a methyl (which turns 3k into 3l and turns 107d into 107e) might
reduce the tendency of the compound to be metabolised. However the team would
regard both the hydroxy and methoxy compounds as having possible sites of
metabolism, albeit without any data their guess would be that hydroxy would be
metabolised more readily than methoxy.
199.
I will not chase down every example of this sort of argument in the
case. They are all instances of a general difficulty. The problem is
concerned with the interpretation of negative results. Accord submitted that
a negative data point could easily be caused by ADME properties, and have
nothing to do with the binding to whatever target the team believed was
relevant. I agree. Accord then went on to submit that:
“as such, negative data does not necessarily prove anything
about the active site, and certainly cannot invalidate a hypothesis that is
built upon positive data. The negative data must be given much less weight,
although the quantity of negative data could ultimately require refinement of
the SAR hypothesis, if sufficiently overwhelming.”
200.
This submission was supported by a contention by Accord that Professor
Cox and Professor Ward were fundamentally at odds about this point and that
that was due to their different experience in work based on animal models (Cox)
as opposed to in vitro assays (Ward). I have already rejected the latter point
about their difference in experience.
201.
As for the former point I do not believe the experts were as far apart
as Accord’s submission suggests. That is because as far as I am aware
Professor Cox did not say that “negative data … certainly cannot invalidate
a hypothesis that is built upon positive data” and I would not have
accepted it if he had. A hypothesis which cannot be falsified by negative data
is not a scientific hypothesis at all and I am sure Professor Cox understood
that as all scientists do. The point Professor Cox was making is best seen in
a passage in cross-examination from Day 3 p337 line 10 to 341 line 18. He
accepted that it was standard medicinal chemistry to build hypotheses based on
active and inactive compounds but also explained that the team would not put
all its energy into explaining why inactive compounds do not work. That is
because their focus would be the active compounds. I accept that. It does not
mean that negative data “is given much less weight” nor does it mean that the
quantity of negative data has to be “overwhelming” to justify changing the SAR
hypothesis. What is the case, as Professor Cox’s evidence explained, was that
the weight of evidence does matter. A body of results pointing one way or
another will be given more weight than a few results.
202.
In the end the issue comes down to the following. As I have said before
Professor Ward’s opinion was essentially that looking at the results as a
whole, positive and negative, the skilled team would not predict that an
aliphatic compound such as 107e, albeit with a β-heteroatom, would have
good activity. That was for two fundamental reasons. First because so much of
the data related to aromatic rather than aliphatic compounds and second because
he thought that while ADME and other effects might be the explanation for the
negative results, then again they might not and in any case the same effects
might just as well apply to compound 107e. To the contrary Professor Cox’s
opinion was that the β-heteroatom theory was supported by a lot of
evidence and where it is not supported there were very realistic ways in which
you can explain why the negative result does not fit.
203.
In my judgment a critical factor is a point emphasised by Professor
Ward, that so much of the Kohn group’s work is on aromatic rather than aliphatic
compounds. Much of the evidence which supports the β-heteroatom theory is
in aromatic compounds. I am not convinced the skilled team, acting without
invention, would be comfortable making predictions about the behaviour of an
aliphatic compound with a β-heteroatom (such as compound 107e) based on
reasoning derived from results achieved using aromatic compounds, even if those
aromatic compounds also had a β-heteroatom. Making predictions based on
comparing aromatic and aliphatic compounds does not compare like with like.
The team considering compound 107e would focus primarily on evidence relating
to the β-heteroatom theory in aliphatic compounds alone. Taking that
approach the weight of positive evidence for β-heteroatomic aliphatic compounds
to set against the negative results for β-heteroatomic aliphatic compounds
makes the position much more equivocal. I find that the team would not think
there was sufficient evidence from which they could make a reasonable inference
that 107e would give good activity in whichever in vivo test they were going to
use.
204.
Although I have focussed on the β-heteroatom theory, it is right to
point out that there were other ways of making the same essential point.
Professor Cox also referred to the need for electron richness in this location
instead of focussing on the β-heteroatom theory. In my judgment this
alternative approach comes to the same point in the end, because in compound
107e the electron richness would be provided by the lone pairs on the oxygen.
If the electron rich approach is not the same as the β-heteroatom approach
then I do not accept that the electron rich way of thinking would justify a
finding of obviousness if the arguments based on the β-heteroatom approach
fails. That is because discussion about a β-heteroatom is the way in
which the authors of the Kohn papers express themselves and is the way the
skilled team acting without invention would think. If electron richness is
different (e.g. because it would be a way of trying to pull together aromatic
and aliphatic results) then I was not satisfied it was an obvious way to
reinterpret the information available to the skilled team.
205.
Putting it all another way, there are just too many uncertainties to
justify a finding of obviousness. Even if the team got as far as deciding to
make and test 107e they would know that the uncertainties meant that either
outcome, good or bad, could just as easily be rationalised after the event as
the other. That is not a fair prospect of success.
206.
So I reject the obviousness case over Le Gall either alone or with the
supplementary information on the primary ground that I am not satisfied that
even if the skilled team did decide to try the relevant tests (using the 107e
racemate in MES and neurotoxicity tests ip in mice or rats), they would have a
sufficient prospect of success to lead to the conclusion that the claim lacks
inventive step.
207.
The arguments delved into further points of detail on a number of issues
– such as: how much the skilled team would know about the results of the NIH
compound testing programme, toxic furan metabolites and ease of synthesis of
enantiomers, what the team might make of the effect of basicity and Kohn 1994,
but I have not found it necessary to decide those questions.
208.
Having reached that conclusion I will also say that I was doubtful that
the whole step by step analysis was indicative of obviousness. The series of
steps from Le Gall involved a full literature search, the distillation of all
that work, numerous other avenues which could be considered and the conception
of the methoxymethyl compound as something to consider. Although they are all
apparently easy steps and many of them are conventional individually, looked at
as a whole they did not strike me as supportive of obviousness but I did not
decide the case on that point.
Bardel
209.
Accord’s alternative case was over Bardel plus supplementary
information. This time the supplementary information relied on is the express
references in the paper. I agree it would be conventional for a team given
Bardel to acquire and read those further papers. They are Kohn 1990, Kohn 1991
and Kohn 1993. I am bound to say on the evidence I would have thought the team
would do essentially the same full literature search as over Le Gall (because I
really doubt such a literature search in 1996 would happen just because of the
age of Le Gall) but this turned out to be contentious and Accord submitted it
was not established. I think Accord’s forensic objective was that this
approach allowed Accord to say that the skilled team did not have the Leander
abstract or Kohn 1994 before them when they make the relevant decisions. The
Leander abstract might inconveniently send the team off an alternative avenue
of chasing LY 274959 while Kohn 1994 might suggest than the nitrogen in
compound 3l was important and should not be replaced. Neither issue has been
part of my reasoning and so the literature search point does not matter and I
will not decide it.
210.
Given the prominence of the Bardel paper in the case advanced over Le
Gall, there is no material difference between the case I have rejected and the
one starting from Bardel. The biggest difference is a point which makes the
case more difficult for Accord because the team would not have Le Gall. At
least starting from Le Gall the team would have seen compound 107e at one stage
even if they had forgotten it after completing and distilling the literature
search. Over Bardel the team would have to think of 107e with no prior contact
with the compound at all.
211.
In any case my findings over Le Gall mean the claims are not obvious
over Bardel either.
Conclusion
212.
The priority date for the relevant claims in the patent is 15th
March 1996. The claims of the patent involve an inventive step and are not
obvious. The action will be dismissed.



 2-pyrrole
(69b):
2-pyrrole
(69b):
 3-
thienyl (69d):
3-
thienyl (69d): 
 2-benzofuryl
(69f):
2-benzofuryl
(69f): 


![]() Ethoxy
(86b)
Ethoxy
(86b) 
 Iso-propyl
(68d)
Iso-propyl
(68d) ![]()
![]()


![]()